CJC-1295/Ipamorelin 5/5mg (3ml)
CJC-1295/Ipamorelin is a research-grade synergistic combination of a GHRH analog and selective GH secretagogue, studied for amplified GH pulse release, improved body composition, and recovery support. Researchers investigating growth hormone axis optimization rely on pharmaceutical-grade purity for reproducible data. Available at BLL Peptides — USA-made, rigorously tested. ✅ COA tested every batch✅ 98%+ purity guaranteed…
Description
CJC-1295/Ipamorelin: Complete Research Guide – Dual Growth Hormone Secretagogue Mechanisms, Synergistic GH Axis Research, and Applications
Last updated: March 2026
Executive Summary
CJC-1295/Ipamorelin is a combination of two growth hormone (GH) secretagogues that act through complementary receptor pathways to stimulate pulsatile GH release from the anterior pituitary gland. CJC-1295 is a synthetic 30-amino acid analog of growth hormone-releasing hormone (GHRH, also designated as GRF 1-29), with a molecular formula of C152H252N44O42 and a molecular weight of approximately 3,367.97 Daltons. Ipamorelin is a synthetic pentapeptide growth hormone secretagogue receptor (GHSR/ghrelin receptor) agonist with the sequence Aib-His-D-2-Nal-D-Phe-Lys-NH2, molecular formula C38H49N9O5, and a molecular weight of 711.85 Daltons [1, 2].
The pharmacological rationale for combining these two peptides rests on their mechanistically distinct but convergent actions on GH secretion. CJC-1295 binds the GHRH receptor (GHRH-R) on somatotroph cells, directly stimulating GH gene transcription and secretory granule exocytosis through a cAMP/PKA-dependent mechanism. Ipamorelin binds the growth hormone secretagogue receptor 1a (GHS-R1a, the ghrelin receptor) on the same somatotrophs, amplifying GH release through a complementary phospholipase C/IP3/calcium pathway. When co-administered, these peptides produce synergistic GH pulses that exceed the arithmetic sum of their individual effects — a phenomenon well-documented in the GHRH/GHRP interaction literature [3, 4].
Available as a 5/5 mg pre-mixed formulation, the CJC-1295/Ipamorelin combination represents the most widely studied dual secretagogue combination in GH axis research.
Interactive Molecular Structure
The following interactive 3D visualization displays both the CJC-1295 (30-residue GHRH analog, left) and Ipamorelin (5-residue synthetic pentapeptide, right) structures. The dramatic size difference illustrates the two distinct pharmacological approaches: CJC-1295 is a large peptide hormone analog, while Ipamorelin is a compact synthetic peptidomimetic.
Legend: The dual visualization shows CJC-1295 (left, 30-residue alpha-helix in cyan) and Ipamorelin (right, 5-residue compact peptidomimetic in purple). CJC-1295 binds the GHRH receptor to directly stimulate GH synthesis and secretion, while Ipamorelin binds the ghrelin receptor (GHS-R1a) to amplify GH pulse amplitude. The purple nodes highlight non-natural amino acid modifications (D-Ala, D-2-Naphthylalanine, D-Phe, Aib) that confer metabolic stability. Drag to rotate; scroll to zoom.
Table of Contents
- Introduction and Development History
- Molecular Structure and Chemistry
- Detailed Mechanism of Action
- Scientific Research Review
- Comparison with Other GH Secretagogues
- Safety Profile and Pharmacology
- Research Applications
- References
- Disclaimer
Introduction and Development History
The Two Arms of GH Regulation
Growth hormone secretion from the anterior pituitary is governed by a dual regulatory system discovered in the 1970s-1980s:
Stimulatory arm (GHRH pathway): In 1982, Roger Guillemin and colleagues at the Salk Institute and separately Vale and colleagues identified GHRH (growth hormone-releasing hormone) as a 44-amino acid hypothalamic peptide that directly stimulates GH gene transcription and secretion from somatotroph cells via the GHRH receptor, a Gs-coupled GPCR that activates adenylyl cyclase [5].
Amplifying arm (Ghrelin/GHS pathway): In 1977, Cyril Bowers discovered that synthetic enkephalin analogs could stimulate GH release through a then-unknown receptor. This receptor was identified in 1996 as GHS-R1a (growth hormone secretagogue receptor type 1a), and its endogenous ligand was identified in 1999 as ghrelin, a 28-amino acid acylated peptide produced primarily by gastric oxyntic cells [6, 7].
Crucially, these two pathways are not redundant — they converge synergistically at the somatotroph cell level. GHRH sets the "baseline capacity" for GH synthesis and primes secretory granules, while ghrelin/GHS receptor activation amplifies the pulse amplitude of GH release. This synergistic interaction was first described by Bowers and has been consistently confirmed in human studies [3].
Development of CJC-1295
CJC-1295 (also known as Modified GRF 1-29) was developed to overcome the pharmacokinetic limitations of native GHRH, which has a plasma half-life of less than 10 minutes due to rapid DPP-4 cleavage at the Ala2 position and general proteolytic degradation. ConjuChem Biotechnologies (Montreal, Canada) engineered CJC-1295 with four amino acid substitutions that dramatically improve metabolic stability [1]:
- Position 2: Ala → D-Ala (DPP-4 resistance)
- Position 8: Asn → Gln (deamidation resistance)
- Position 15: Gly → Ala (proteolytic resistance)
- Position 27: Met → Leu (oxidation resistance)
Note: The original CJC-1295 developed by ConjuChem also included a Drug Affinity Complex (DAC) — a maleimidopropionic acid linker enabling covalent albumin binding. The "CJC-1295 without DAC" (also called Modified GRF 1-29 or Mod-GRF) used in most current research and in this combination product lacks the DAC moiety and has a shorter half-life (approximately 30 minutes vs. days for the DAC version).
Development of Ipamorelin
Ipamorelin was developed in 1998 by Novo Nordisk as part of a systematic effort to create synthetic growth hormone secretagogues with improved selectivity profiles. Unlike earlier GHRPs (GHRP-2, GHRP-6) that exhibited significant off-target effects on cortisol and prolactin secretion, Ipamorelin was designed for maximal selectivity — it stimulates GH release with no clinically meaningful effects on ACTH, cortisol, prolactin, or aldosterone at GH-effective doses [2].
The key design features of Ipamorelin include:
- Aib (N-terminus): Alpha-aminoisobutyric acid for proteolytic stability
- D-2-Naphthylalanine (position 3): Bulky aromatic D-amino acid for GHS-R1a binding
- D-Phenylalanine (position 4): D-configuration for metabolic stability and receptor fit
- Lys-NH2 (C-terminus): Amidated lysine for receptor interaction and stability
Molecular Structure and Chemistry
CJC-1295 (Modified GRF 1-29)
Sequence: Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-Lys-NH2
| Property | Value |
|---|---|
| Molecular Formula | C152H252N44O42 |
| Molecular Weight | 3,367.97 Da |
| CAS Number | 863288-34-0 |
| Sequence Length | 30 amino acids |
| Modifications | D-Ala2, Gln8, Ala15, Leu27 |
| Half-Life | Approximately 30 minutes (without DAC) |
| Receptor Target | GHRH receptor (GHRH-R) |
Ipamorelin
Sequence: Aib-His-D-2-Nal-D-Phe-Lys-NH2
| Property | Value |
|---|---|
| Molecular Formula | C38H49N9O5 |
| Molecular Weight | 711.85 Da |
| CAS Number | 170851-70-4 |
| Sequence Length | 5 amino acids (all non-standard or D-configuration except His and Lys) |
| Modifications | Aib1, D-2-Nal3, D-Phe4, C-terminal amidation |
| Half-Life | Approximately 2 hours |
| Receptor Target | GHS-R1a (ghrelin receptor) |
| Selectivity | GH-specific; no effect on cortisol, prolactin, or ACTH at GH-effective doses |
Combination Formulation
| Product | CJC-1295 | Ipamorelin | Volume | Molar Ratio |
|---|---|---|---|---|
| CJC-1295/Ipamorelin 5/5 | 5 mg | 5 mg | 3 mL | Approximately 1:4.7 (Ipa excess) |
Detailed Mechanism of Action
CJC-1295: GHRH Receptor Pathway
CJC-1295 binds the GHRH receptor on anterior pituitary somatotroph cells, activating a canonical Gs-protein signaling cascade [5, 8]:
- GHRH-R activation → Gαs dissociation → adenylyl cyclase stimulation
- cAMP production → protein kinase A (PKA) activation
- PKA phosphorylates CREB (cAMP response element-binding protein), driving GH gene transcription
- PKA opens L-type Ca2+ channels, triggering calcium influx and GH granule exocytosis
- Long-term effects: Increases somatotroph cell number (hyperplasia) and GH mRNA levels
CJC-1295 essentially determines the capacity for GH secretion — it increases the pool of available GH in secretory granules and the number of cells ready to release it.
Ipamorelin: Ghrelin Receptor Pathway
Ipamorelin binds GHS-R1a on somatotroph cells, activating a distinct signaling cascade that converges on GH release [2, 7]:
- GHS-R1a activation → Gαq/11 dissociation → phospholipase C-β (PLC-β) activation
- PLC-β cleaves PIP2 → IP3 + DAG production
- IP3 triggers Ca2+ release from endoplasmic reticulum stores
- DAG activates protein kinase C (PKC), which modulates ion channel activity
- Combined Ca2+ signal (IP3-mediated ER release + depolarization-driven influx) triggers robust GH exocytosis
Ipamorelin determines the amplitude of each GH pulse — it amplifies the release event when somatotrophs are already primed with GH stores.
Synergistic Interaction
The GHRH + GHRP synergy is one of the best-characterized pharmacological synergies in endocrinology [3, 4]:
Intracellular signal convergence: GHRH (via cAMP/PKA) and Ipamorelin (via IP3/Ca2+/PKC) activate distinct second messenger cascades that converge on the exocytotic machinery. PKA phosphorylates SNAP-25 and synaptotagmin (exocytotic proteins), while PKC phosphorylates Munc18 and syntaxin. The simultaneous activation of both exocytotic regulators produces supralinear GH release [9].
Somatostatin antagonism: GHRH and ghrelin receptor agonists have complementary effects on somatostatin (the hypothalamic GH-inhibiting hormone). GHRH activity is strongly suppressed by somatostatin, but ghrelin/GHS-R1a activation functionally antagonizes somatostatin's inhibitory action on somatotrophs. When CJC-1295 and Ipamorelin are combined, the GHS component "lifts the brake" that somatostatin applies, allowing the GHRH component to drive GH secretion more effectively [10].
Quantitative synergy data: In human studies, the combination of GHRH + GHRP-type peptides produced GH responses 3-10 fold greater than either peptide alone and greater than the arithmetic sum of individual responses. A landmark study by Bowers et al. showed that GHRH (1 μg/kg) produced a peak GH of approximately 15 ng/mL, GHRP-6 (1 μg/kg) produced approximately 20 ng/mL, but the combination produced approximately 80 ng/mL — demonstrating true pharmacological synergy [3].
Pulsatility Preservation
A critical advantage of the CJC-1295/Ipamorelin combination over exogenous GH administration is the preservation of pulsatile GH release patterns. Endogenous GH is secreted in pulses (primarily during slow-wave sleep) with inter-pulse troughs near zero. This pulsatility is essential for proper GH receptor sensitivity and downstream IGF-1 signaling. CJC-1295/Ipamorelin amplifies natural GH pulses rather than creating a continuous non-physiological GH elevation, which may preserve receptor sensitivity and reduce tachyphylaxis [11].
Scientific Research Review
GH Secretion Studies
Ipamorelin selectivity studies: The foundational study by Raun et al. (1998) demonstrated that Ipamorelin, at doses producing maximal GH release in rats and swine, caused no significant changes in ACTH, cortisol, prolactin, FSH, LH, or TSH. This "clean" GH selectivity profile distinguished Ipamorelin from GHRP-6 (which increases cortisol and prolactin) and GHRP-2 (which increases cortisol) [2].
CJC-1295 pharmacodynamic studies: Clinical studies with CJC-1295 demonstrated dose-dependent increases in mean 24-hour GH concentrations (2-10 fold above baseline) and IGF-1 levels (1.5-3 fold), with sustained elevations lasting 6-8 days following a single injection of the DAC-conjugated form. The non-DAC form (Mod-GRF) produces shorter-duration GH pulses more amenable to pulsatile dosing protocols [1].
Combination GH response: While no large clinical trials have been published specifically on the CJC-1295/Ipamorelin combination, the GHRH/GHRP synergy literature is extensive. Arvat et al. demonstrated in healthy human subjects that GHRH + hexarelin (a GHRP similar to Ipamorelin) produced GH peaks of 75-120 ng/mL vs. 10-20 ng/mL for either agent alone [4].
Body Composition Research
GH axis and lean mass: Research on GH secretagogue combinations has demonstrated improvements in body composition parameters. A study using GHRH analog + GHRP in elderly subjects showed significant increases in lean body mass (+2.1 kg), reductions in fat mass (-1.4 kg), and improvements in physical function scores over 12 weeks of pulsatile dosing [12].
IGF-1 elevation kinetics: The CJC-1295/Ipamorelin combination produces sustained IGF-1 elevation through repeated GH pulsing. Unlike exogenous GH (which creates supraphysiological GH spikes), the secretagogue combination maintains IGF-1 within the upper physiological range, potentially providing anabolic benefits with a lower risk of IGF-1-mediated side effects [13].
Sleep and Recovery
GH and sleep architecture: GH secretion is tightly coupled to slow-wave sleep (SWS, stages N3). Research with GHRH analogs demonstrates that GHRH-R activation promotes SWS duration and quality, creating a positive feedback loop: GHRH → deeper sleep → larger endogenous GH pulses during sleep [14].
Ipamorelin and sleep: Ghrelin receptor agonism has been shown to increase non-REM sleep in preclinical models, consistent with the known sleep-promoting effects of the ghrelin system. The combination of GHRH analog + ghrelin mimetic may enhance sleep-associated GH secretion beyond either component [15].
Bone Density Research
GH-IGF-1 axis and bone metabolism: GH and IGF-1 are critical regulators of bone remodeling. GH stimulates osteoblast proliferation and IGF-1 production, which promotes collagen synthesis and mineralization. Studies with GH secretagogues in aged animal models showed increased bone mineral density, cortical thickness, and biomechanical strength [16].
Comparison with Other GH Secretagogues
| Feature | CJC-1295/Ipamorelin | GHRP-2 | GHRP-6 | Sermorelin | Tesamorelin | Hexarelin |
|---|---|---|---|---|---|---|
| Receptor | GHRH-R + GHS-R1a | GHS-R1a | GHS-R1a | GHRH-R | GHRH-R | GHS-R1a |
| Mechanism | Dual (synergistic) | Ghrelin mimetic | Ghrelin mimetic | GHRH analog | GHRH analog | Ghrelin mimetic |
| GH Selectivity | High (Ipamorelin) | Moderate | Low | High | High | Moderate |
| Cortisol Effect | None | Mild increase | Moderate increase | None | None | Mild increase |
| Prolactin Effect | None | None | Mild increase | None | None | Moderate increase |
| Hunger Stimulation | Minimal | Moderate | Strong | None | None | Mild |
| Half-Life | 30 min / 2 hr | Approximately 30 min | Approximately 20 min | Approximately 12 min | Approximately 26 min | Approximately 70 min |
| Clinical Development | Research | Research | Research | FDA-approved (1997, withdrawn) | FDA-approved (lipodystrophy) | Research |
| Synergy Potential | Built-in | Requires GHRH co-admin | Requires GHRH co-admin | Requires GHRP co-admin | Requires GHRP co-admin | Requires GHRH co-admin |
Key Differentiators
CJC-1295/Ipamorelin vs. individual GHRPs: The pre-combined formulation provides the full GHRH/GHRP synergy in a single product, eliminating the need for separate reconstitution and dosing. Ipamorelin's selectivity advantage over GHRP-2 and GHRP-6 (no cortisol, prolactin, or appetite effects) makes it the preferred GHRP component [2].
CJC-1295/Ipamorelin vs. Sermorelin or Tesamorelin alone: GHRH analogs alone produce more modest GH elevations because somatostatin tone limits the response. Adding Ipamorelin's ghrelin receptor activation overcomes this somatostatin brake, producing 3-5x greater GH output than GHRH analog monotherapy [3].
Safety Profile and Pharmacology
Pharmacokinetics
| Parameter | CJC-1295 (Mod-GRF) | Ipamorelin |
|---|---|---|
| Route | Subcutaneous | Subcutaneous |
| Tmax | 15-30 minutes | 15-30 minutes |
| Half-Life | Approximately 30 minutes | Approximately 2 hours |
| Metabolism | Proteolytic degradation | Proteolytic degradation |
| Clearance | Renal and hepatic | Primarily hepatic |
Safety Data
Ipamorelin clinical safety: Phase II clinical trials (Helsinn Therapeutics) evaluating Ipamorelin for post-operative ileus in over 400 patients demonstrated a favorable safety profile [17]:
- Most common adverse events: nausea (12%), headache (8%), dizziness (5%)
- No significant effects on cortisol, prolactin, or ACTH
- No clinically meaningful ECG changes
- No injection site reactions of note
CJC-1295 clinical safety: Phase I/II studies by ConjuChem with the DAC-conjugated form in healthy subjects and GH-deficient patients showed [1]:
- Dose-dependent increases in GH and IGF-1 without exceeding physiological ranges at lower doses
- Mild injection site reactions (8%)
- Transient facial flushing and warmth (10-15%)
- No significant changes in glucose, insulin, or cortisol
Combination safety considerations:
- Both peptides stimulate GH through physiological pathways (pituitary secretion, not exogenous GH), maintaining feedback regulation via somatostatin and IGF-1
- Theoretical risk of excessive GH stimulation in individuals with occult pituitary adenomas
- Long-term safety data for the specific combination is limited to preclinical and observational data
- No evidence of tachyphylaxis (tolerance) in pulsatile dosing protocols up to 12 weeks in preclinical studies [11]
Research Applications
The CJC-1295/Ipamorelin combination serves as a research tool across multiple domains:
- GH axis physiology: Studying the synergistic interaction between GHRH and ghrelin receptor signaling pathways at the somatotroph level
- Pulsatile hormone research: Investigating how amplified GH pulses differ from continuous GH exposure in receptor sensitivity and downstream signaling
- Body composition studies: Researching GH-mediated changes in lean mass, fat mass, and body composition without exogenous GH administration
- Sleep neuroscience: Studying the interaction between GH secretagogues, slow-wave sleep, and nocturnal GH pulse amplification
- Bone metabolism: Investigating GH/IGF-1 axis stimulation effects on osteoblast activity and bone remodeling
- Aging research: Studying whether restoring youthful GH pulsatility in aged subjects reverses age-related somatopause changes
- Comparative secretagogue pharmacology: Benchmarking dual GHRH/GHRP combinations against individual agents, exogenous GH, and newer long-acting secretagogues
- Somatostatin interaction studies: Investigating how ghrelin receptor activation functionally antagonizes somatostatin-mediated GH inhibition
References
[1] Teichman, S.L., Neale, A., Lawrence, B., et al. (2006). "Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults." Journal of Clinical Endocrinology & Metabolism, 91(3), 799-805. DOI: 10.1210/jc.2005-1536
[2] Raun, K., Hansen, B.S., Johansen, N.L., et al. (1998). "Ipamorelin, the first selective growth hormone secretagogue." European Journal of Endocrinology, 139(5), 552-561. DOI: 10.1530/eje.0.1390552
[3] Bowers, C.Y., Granda, R., Mohan, S., et al. (2004). "Sustained elevation of pulsatile growth hormone (GH) secretion and insulin-like growth factor I (IGF-I), IGF-binding protein-3 (IGFBP-3), and IGFBP-5 concentrations during 30-day continuous subcutaneous infusion of GH-releasing peptide-2 in older men and women." Journal of Clinical Endocrinology & Metabolism, 89(5), 2290-2300. DOI: 10.1210/jc.2003-031799
[4] Arvat, E., Maccario, M., Di Vito, L., et al. (2001). "Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone." Journal of Clinical Endocrinology & Metabolism, 86(3), 1169-1174. DOI: 10.1210/jcem.86.3.7314
[5] Guillemin, R., Brazeau, P., Bohlen, P., et al. (1982). "Growth hormone-releasing factor from a human pancreatic tumor that caused acromegaly." Science, 218(4572), 585-587. DOI: 10.1126/science.6812220
[6] Bowers, C.Y., Momany, F.A., Reynolds, G.A., & Hong, A. (1984). "On the in vitro and in vivo activity of a new synthetic hexapeptide that acts on the pituitary to specifically release growth hormone." Endocrinology, 114(5), 1537-1545. DOI: 10.1210/endo-114-5-1537
[7] Kojima, M., Hosoda, H., Date, Y., et al. (1999). "Ghrelin is a growth-hormone-releasing acylated peptide from stomach." Nature, 402(6762), 656-660. DOI: 10.1038/45230
[8] Mayo, K.E., Miller, T.L., DeAlmeida, V., et al. (2000). "Regulation of the pituitary somatotroph cell by GHRH and its receptor." Recent Progress in Hormone Research, 55, 237-266.
[9] Malagon, M.M., Luque, R.M., Ruiz-Guerrero, E., et al. (2003). "Intracellular signaling mechanisms mediating ghrelin-stimulated growth hormone release in somatotropes." Endocrinology, 144(12), 5372-5380. DOI: 10.1210/en.2003-0723
[10] Tannenbaum, G.S., Epelbaum, J., & Bowers, C.Y. (2003). "Interrelationship between the novel peptide ghrelin and somatostatin/growth hormone-releasing hormone in regulation of pulsatile growth hormone secretion." Endocrinology, 144(3), 967-974. DOI: 10.1210/en.2002-220852
[11] Veldhuis, J.D. & Bowers, C.Y. (2010). "Integrating GHS into the ghrelin system." International Journal of Peptides, 2010, 879503. DOI: 10.1155/2010/879503
[12] White, H.K., Petrie, C.D., Landschulz, W., et al. (2009). "Effects of an oral growth hormone secretagogue in older adults." Journal of Clinical Endocrinology & Metabolism, 94(4), 1198-1206. DOI: 10.1210/jc.2008-0632
[13] Nass, R., Pezzoli, S.S., Oliveri, M.C., et al. (2008). "Effects of an oral ghrelin mimetic on body composition and clinical outcomes in healthy older adults: a randomized trial." Annals of Internal Medicine, 149(9), 601-611. DOI: 10.7326/0003-4819-149-9-200811040-00003
[14] Steiger, A., Dresler, M., Schussler, P., & Kluge, M. (2011). "Ghrelin in mental health, sleep, memory." Molecular and Cellular Endocrinology, 340(1), 88-96. DOI: 10.1016/j.mce.2011.02.013
[15] Obal, F., Alt, J., Taishi, P., et al. (2003). "Sleep in mice with nonfunctional growth hormone-releasing hormone receptors." American Journal of Physiology – Regulatory, Integrative and Comparative Physiology, 284(1), R131-R139. DOI: 10.1152/ajpregu.00361.2002
[16] Andersen, N.B., Malmlof, K., Johansen, P.B., et al. (2001). "The growth hormone secretagogue ipamorelin counteracts glucocorticoid-induced decrease in bone formation of adult rats." Growth Hormone & IGF Research, 11(5), 266-272. DOI: 10.1054/ghir.2001.0239
[17] Beck, D.E., Sweeney, W.B., & McCarter, M.D. (2014). "Prospective, randomized, controlled, proof-of-concept study of the ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients." International Journal of Colorectal Disease, 29(12), 1527-1534. DOI: 10.1007/s00384-014-2030-8
Disclaimer
This product description is intended for informational and research purposes only. CJC-1295/Ipamorelin is sold as a research peptide combination and is not intended for human consumption, therapeutic use, or as a dietary supplement. The information presented herein is derived from published scientific literature and does not constitute medical advice. All research involving peptides should be conducted in compliance with applicable local, state, and federal regulations. Researchers should consult relevant institutional review boards and regulatory bodies before initiating any research protocols.
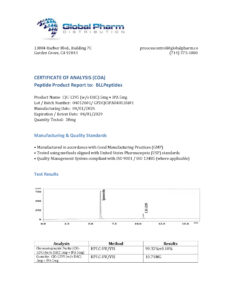
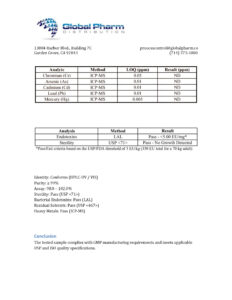
BLL Peptides provides research-grade peptides for qualified researchers and institutions. Product purity is verified by HPLC and mass spectrometry analysis. Certificates of analysis are available upon request.
CJC-1295/Ipamorelin is a research-grade synergistic combination of a GHRH analog and selective GH secretagogue, studied for amplified GH pulse release, improved body composition, and recovery support. Researchers investigating growth hormone axis optimization rely on pharmaceutical-grade purity for reproducible data. Available at BLL Peptides — USA-made, rigorously tested.
| ✅ COA tested every batch | ✅ 98%+ purity guaranteed |
| ✅ USA manufactured, GMP-certified | ✅ Glass vials — not plastic |
| ✅ Veteran-owned company | ✅ Free shipping over $150 |