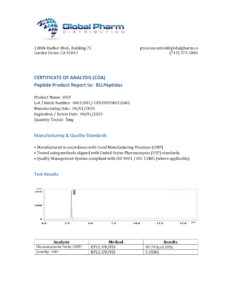
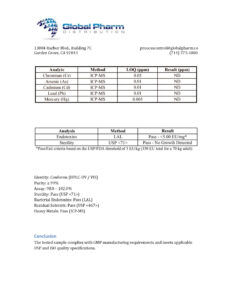
DSIP 10mg (3ml)
DSIP (Delta Sleep-Inducing Peptide) is a research-grade neuropeptide studied for roles in sleep architecture regulation, stress modulation, neuroendocrine function, and antioxidant activity. Researchers investigating sleep biology, circadian rhythm mechanisms, and neuropeptide pharmacology rely on pharmaceutical-grade purity for accurate results. Available at BLL Peptides — USA-made, rigorously tested. ✅ COA tested every batch✅ 98%+ purity guaranteed…
Description
DSIP: Complete Research Guide – Delta Sleep-Inducing Peptide Mechanisms, Sleep Architecture Research, and Neuroendocrine Applications
Last updated: March 2026
Executive Summary
Delta Sleep-Inducing Peptide (DSIP) is a naturally occurring nonapeptide with the amino acid sequence Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu (WAGGDASGE). With a molecular formula of C35H48N10O15 and a molecular weight of approximately 848.81 Daltons (CAS: 62568-57-4), DSIP was first isolated in 1977 by Schoenenberger and Monnier from the cerebral venous blood of rabbits during electrically induced slow-wave sleep [1]. The peptide was identified through a landmark cross-perfusion experiment in which venous blood from sleeping donor rabbits was infused into the cerebral circulation of alert recipient rabbits, inducing characteristic delta-wave electroencephalographic (EEG) patterns in the recipients.
DSIP's discovery marked a watershed moment in sleep neurochemistry, providing the first direct peptidergic evidence that sleep-promoting substances circulate in the bloodstream. Unlike classical neurotransmitter-based sleep modulators such as GABA or adenosine, DSIP represents a humoral, peptidergic factor capable of crossing the blood-brain barrier via a non-saturable mechanism that is partially independent of conventional receptor-mediated transport [2, 3]. This unique pharmacokinetic property enables peripheral administration to produce central nervous system effects, a characteristic of considerable research significance.
The biological activity of DSIP extends far beyond its original characterization as a sleep factor. Subsequent decades of research have revealed that DSIP modulates the hypothalamic-pituitary-adrenal (HPA) axis, suppressing stress-induced elevations in adrenocorticotropic hormone (ACTH) and cortisol [4]. It demonstrates significant antioxidant properties through the induction of endogenous antioxidant enzyme systems [5], exhibits analgesic activity in multiple pain models [6], and has shown promise in attenuating withdrawal symptoms in opioid-dependent subjects [7]. DSIP also influences circadian rhythm regulation, interacting with the suprachiasmatic nucleus and modulating melatonin secretion [8].
This comprehensive research guide examines the molecular structure, mechanisms of action, published research findings, safety profile, and investigational applications of DSIP. For researchers exploring related neuropeptides and neuromodulatory compounds, see also our guides on Selank, Pinealon, and Epithalon.
Interactive Molecular Structure
The following interactive 3D visualization renders the DSIP nonapeptide (Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu) in an extended beta-turn conformation. As a glycine-rich peptide (three of nine residues are glycine), DSIP possesses exceptional backbone flexibility and cannot adopt stable alpha-helical or beta-sheet structures. The N-terminal tryptophan (cyan) serves as the aromatic anchor, while the two acidic residues, Asp5 and Glu9 (orange), confer the peptide's overall negative charge at physiological pH. The small glycine residues (silver-blue) provide the conformational flexibility that is critical for DSIP's ability to interact with multiple receptor systems.
Table of Contents
- Introduction and Historical Context
- Molecular Structure and Chemistry
- Mechanism of Action
- Scientific Research Review
- Comparison with Related Neuroactive Peptides
- Safety Profile and Tolerability
- Research Applications and Investigational Directions
- References
- Disclaimer
Introduction and Historical Context
The Discovery of a Sleep Factor
The story of DSIP begins with one of the most elegant experiments in sleep neuroscience. In 1977, Swiss researchers GA Schoenenberger and M Monnier at the University of Basel performed cross-circulation experiments in rabbits, in which they stimulated the intralaminar thalamic nuclei of donor rabbits to induce slow-wave sleep while simultaneously collecting cerebral venous blood [1]. When the dialyzed blood fraction from these sleeping donors was infused into the cerebral ventricles of alert recipient rabbits, the recipients exhibited a significant increase in delta-wave (0.5 to 4 Hz) EEG activity characteristic of deep sleep stages. The active substance was subsequently purified and identified as a nonapeptide, which the researchers named Delta Sleep-Inducing Peptide.
The isolation and sequencing of DSIP was accomplished through successive chromatographic fractionation of the cerebral venous blood dialysate. The peptide was ultimately sequenced as Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu using Edman degradation, and its identity was confirmed by chemical synthesis [1, 9]. This work represented a major advance in the emerging field of sleep neurochemistry, which was still dominated by the "humoral theory of sleep" first proposed by Ishimori (1909) and Legendre and Pieron (1913), who had demonstrated that cerebrospinal fluid from sleep-deprived dogs could induce sleep in alert recipients.
Evolution of Understanding
The initial characterization of DSIP as purely a "sleep peptide" proved to be an oversimplification. Throughout the 1980s and 1990s, research laboratories worldwide reported an unexpectedly diverse range of biological activities. Graf and Kastin (1984) demonstrated that DSIP could cross the blood-brain barrier in mice through a non-saturable mechanism [2], establishing that peripheral administration could produce central effects. Sudakov et al. (1983) reported stress-protective effects in animal models [10], and Bjartell et al. (1990) used immunohistochemistry to map DSIP-like immunoreactivity to the hypothalamus, limbic system, pituitary gland, and peripheral endocrine tissues, suggesting roles far beyond sleep regulation [11].
A significant turning point came with the work of Kovalzon and Strekalova (2006), who critically reviewed three decades of DSIP research and argued that the peptide should be reconceptualized not as a specific sleep-inducing factor but rather as a stress-protective and homeostatic modulator that promotes sleep as one component of its broader adaptive function [12]. This reinterpretation aligns with the observation that DSIP does not reliably induce sleep in all experimental paradigms but consistently demonstrates protective effects against various forms of physiological stress.
Distribution and Endogenous Sources
DSIP-like immunoreactivity has been detected in numerous tissues and body fluids across mammalian species. In the central nervous system, the highest concentrations are found in the hypothalamus (particularly the arcuate nucleus and median eminence), the limbic structures, and the pituitary gland [11]. Peripherally, DSIP-like immunoreactivity has been identified in the adrenal glands, gastrointestinal tract, and circulating plasma, where concentrations exhibit circadian variation with peak levels occurring during nighttime hours [13]. The peptide has also been detected in human breast milk and cerebrospinal fluid, suggesting both central and peripheral physiological roles [11].
Despite decades of research, a specific membrane receptor for DSIP has not been conclusively identified. This absence of a defined receptor, combined with DSIP's remarkably broad spectrum of biological activities, has led some investigators to propose that DSIP may function not through a single receptor-mediated pathway but rather through modulation of multiple signaling systems simultaneously, potentially acting as an allosteric modulator or membrane-active peptide [12, 14].
Molecular Structure and Chemistry
Primary Structure and Physicochemical Properties
DSIP is a linear nonapeptide consisting of nine amino acid residues in the sequence: H-Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu-OH (single-letter code: WAGGDASGE). Its molecular formula is C35H48N10O15, corresponding to a molecular weight of approximately 848.81 Da. The peptide carries a net negative charge at physiological pH (approximately 7.4) due to the two acidic residues, Asp5 (pKa approximately 3.65) and Glu9 (pKa approximately 4.25), which are deprotonated at neutral pH. The isoelectric point (pI) of DSIP is calculated at approximately 3.47, making it one of the most acidic neuropeptides known [1, 15].
| Property | Value |
|---|---|
| Amino acid sequence | Trp-Ala-Gly-Gly-Asp-Ala-Ser-Gly-Glu |
| One-letter code | WAGGDASGE |
| Molecular formula | C35H48N10O15 |
| Molecular weight | Approximately 848.81 Da |
| CAS number | 62568-57-4 |
| Net charge at pH 7.4 | -2 |
| Isoelectric point (pI) | Approximately 3.47 |
| Number of residues | 9 (nonapeptide) |
| Appearance | White to off-white lyophilized powder |
| Solubility | Freely soluble in water, aqueous buffers |
| Storage | -20 C, desiccated, protected from light |
Conformational Analysis
The three-dimensional structure of DSIP in solution has been investigated using circular dichroism (CD) spectroscopy and nuclear magnetic resonance (NMR). The high glycine content (three of nine residues, or 33%) confers exceptional backbone flexibility. Glycine, lacking a side chain entirely, imposes no steric restrictions on the phi and psi dihedral angles of the peptide backbone, meaning that DSIP can sample a broad conformational ensemble rather than adopting a single stable fold [15].
CD spectroscopy studies indicate that DSIP in aqueous solution exists predominantly in an unordered (random coil) conformation, with some population of beta-turn structures centered around the Gly3-Gly4 dipeptide [15]. This is consistent with the general principle that short peptides (fewer than approximately 15-20 residues) typically cannot form stable secondary structures in the absence of stabilizing constraints such as disulfide bonds, cyclization, or interactions with binding partners.
The N-terminal tryptophan residue deserves particular structural attention. Tryptophan possesses the largest aromatic side chain among the natural amino acids, featuring a bicyclic indole ring system. In DSIP, the indole ring of Trp1 may engage in cation-pi interactions with positively charged residues on binding partners, or in aromatic stacking interactions with membrane lipids, potentially contributing to DSIP's ability to interact with and traverse biological membranes [2]. The intrinsic fluorescence of the tryptophan indole (excitation approximately 280 nm, emission approximately 340 nm) also provides a convenient spectroscopic handle for monitoring DSIP binding interactions and conformational changes.
Stability and Degradation
DSIP is susceptible to enzymatic degradation by both aminopeptidases and carboxypeptidases. In plasma, the peptide has a relatively short half-life, estimated at approximately 10 to 15 minutes in rodent studies, though some reports suggest that DSIP may associate with carrier proteins in the bloodstream that partially protect it from degradation [16]. The Trp1-Ala2 peptide bond at the N-terminus is a primary cleavage site for aminopeptidases, while the C-terminal Glu9 is targeted by carboxypeptidases.
To address the stability limitation, several structural analogs have been developed. The most widely studied is DSIP phosphorylated at the Ser7 hydroxyl group (p-DSIP or DSIP-P), which demonstrates enhanced metabolic stability and, in some studies, augmented biological activity relative to the native peptide [17]. The phosphoserine modification likely confers resistance to serine-targeting proteases while also introducing an additional negative charge that may influence receptor interactions and membrane permeability. Other modifications explored in the literature include D-amino acid substitutions, N-terminal acetylation, and C-terminal amidation, each producing analogs with distinct pharmacological profiles [15].
Mechanism of Action
Blood-Brain Barrier Penetration
One of DSIP's most pharmacologically significant properties is its ability to cross the blood-brain barrier (BBB). Graf and Kastin (1984) demonstrated using radiolabeled DSIP that the peptide enters the brain from peripheral circulation via a non-saturable, partially enzymatically mediated mechanism [2]. This transport does not follow classical receptor-mediated transcytosis (which would show saturation kinetics) but appears to involve a combination of passive diffusion through lipophilic domains of the BBB endothelium and adsorptive-mediated transcytosis facilitated by the peptide's amphipathic character [3].
Banks et al. (1986) further characterized DSIP's BBB transport and found that the peptide's entry into the brain is modified by several factors, including aluminum exposure and sleep deprivation, suggesting that the BBB permeability to DSIP is physiologically regulated [3]. The amphipathic nature of DSIP, with its hydrophobic N-terminus (Trp-Ala) and hydrophilic, negatively charged C-terminus (Gly-Glu), likely enables interaction with the lipid bilayer of the BBB endothelium while maintaining sufficient aqueous solubility for distribution through the plasma.
Sleep Architecture Modulation
DSIP's effects on sleep are mediated through modulation of sleep architecture rather than simple sedation. Electroencephalographic studies in both animal models and human subjects have demonstrated that DSIP preferentially enhances slow-wave sleep (SWS, stages N3) characterized by high-amplitude delta waves (0.5 to 4 Hz), while generally preserving or normalizing rapid eye movement (REM) sleep proportions [1, 18].
The neurochemical basis of DSIP's sleep-promoting effects involves multiple parallel mechanisms:
GABAergic modulation: DSIP has been shown to potentiate GABAergic transmission in hypothalamic sleep-promoting nuclei, including the ventrolateral preoptic area (VLPO). This potentiation may involve enhanced GABA release, upregulation of GABAA receptor sensitivity, or both. The VLPO is a critical node in the sleep-wake flip-flop switch model, and enhanced GABAergic output from the VLPO inhibits wake-promoting centers in the lateral hypothalamus (orexin neurons) and the tuberomammillary nucleus (histamine neurons) [18, 19].
Serotonergic interactions: Research has demonstrated that DSIP modulates serotonin (5-HT) turnover in the raphe nuclei, which play a central role in sleep-wake transitions. Specifically, DSIP appears to facilitate the decline in serotonergic firing that normally accompanies the transition from wakefulness to non-REM sleep [19].
Glutamatergic buffering: Evidence suggests that DSIP may attenuate excitatory glutamatergic neurotransmission in cortical and subcortical arousal circuits, contributing to the reduction in cortical activation that permits delta-wave generation during deep sleep [12].
Hypothalamic-Pituitary-Adrenal (HPA) Axis Regulation
DSIP exerts significant modulatory effects on the HPA axis, the body's primary stress response system. Multiple studies have demonstrated that DSIP administration suppresses stress-induced elevations in ACTH and cortisol, both in animal models and in human subjects [4, 20].
The mechanism of HPA axis modulation appears to involve direct action at the hypothalamic level. DSIP-like immunoreactivity is concentrated in the paraventricular nucleus (PVN) of the hypothalamus, the primary source of corticotropin-releasing hormone (CRH). Evidence suggests that DSIP may attenuate CRH release from PVN neurons, thereby reducing downstream ACTH secretion from the anterior pituitary and, consequently, cortisol release from the adrenal cortex [4, 11].
Importantly, DSIP does not appear to suppress basal HPA axis activity but rather selectively attenuates stress-induced activation, suggesting a modulatory rather than inhibitory mechanism. This selectivity is pharmacologically significant because it implies that DSIP would not produce the adrenal insufficiency associated with chronic glucocorticoid suppression but rather restore stress-axis homeostasis [4, 20].
Antioxidant Properties
DSIP demonstrates notable antioxidant activity that extends beyond direct radical scavenging. Research by Bondarenko (2004) and colleagues has shown that DSIP administration upregulates the expression and activity of endogenous antioxidant enzymes, including superoxide dismutase (SOD), catalase, and glutathione peroxidase, in multiple tissues [5]. This indirect antioxidant mechanism is particularly significant because it amplifies the body's own defense systems rather than merely providing stoichiometric radical scavenging.
The antioxidant effects of DSIP may be mechanistically linked to its stress-protective properties. Chronic stress and HPA axis hyperactivation are known to promote oxidative stress through multiple pathways, including mitochondrial dysfunction, NADPH oxidase activation, and depletion of reduced glutathione. By attenuating HPA axis hyperactivation, DSIP may secondarily reduce oxidative stress burden [5]. For researchers interested in antioxidant peptides and their applications, the glutathione system represents another critical pathway of investigation.
Opioidergic Interactions
DSIP demonstrates complex interactions with the endogenous opioid system. While DSIP itself is not an opioid peptide and does not bind directly to classical opioid receptors (mu, delta, kappa) at physiologically relevant concentrations, it modulates opioidergic signaling through indirect mechanisms [6, 7]. Studies have shown that DSIP can modify the release and metabolism of endogenous opioid peptides, including beta-endorphin and met-enkephalin, potentially through hypothalamic regulatory circuits [6].
This opioidergic modulation may underlie DSIP's reported analgesic effects and its ability to attenuate opioid withdrawal symptoms. In animal models, DSIP administration reduces the severity of naloxone-precipitated withdrawal in morphine-dependent subjects, suggesting that it may stabilize opioidergic neurotransmission during the acute withdrawal period [7].
Scientific Research Review
Sleep Research
Original Discovery and Replication Studies
The foundational work of Schoenenberger and Monnier (1977) demonstrated that synthetic DSIP, when administered intravenously or intracerebroventricularly to rabbits, increased the proportion of delta-wave EEG activity by approximately 28% relative to controls during the first two hours after injection [1]. Subsequent studies by the same group confirmed dose-dependent delta wave enhancement, with optimal effects observed at doses of 10 to 30 nmol/kg intravenously.
However, replication of DSIP's sleep-promoting effects has been inconsistent across laboratories and experimental paradigms. Some groups reported robust delta wave enhancement [18], while others found minimal or no effect on sleep parameters [12]. Kovalzon and Strekalova (2006) comprehensively reviewed these discrepancies and concluded that the variability may reflect differences in animal strain, circadian timing of administration, stress state of the subjects, and the purity of DSIP preparations used [12]. They proposed that DSIP's sleep-promoting effects are most reliably observed in stressed or sleep-disturbed animals, rather than in undisturbed subjects with normal sleep patterns.
Human Sleep Studies
Clinical investigations of DSIP in human subjects have yielded promising but variable results. Schneider-Helmert and Schoenenberger (1983) conducted a controlled study in patients with chronic insomnia and found that repeated intravenous DSIP administration (25 nmol/kg for five consecutive evenings) significantly improved subjective sleep quality and increased total sleep time, with effects persisting for several weeks after the final injection [18]. Polysomnographic analysis revealed a selective increase in slow-wave sleep without suppression of REM sleep, a profile distinctly different from that of benzodiazepine hypnotics, which typically suppress both SWS and REM sleep.
A subsequent study by Schneider-Helmert (1985) in patients with insomnia associated with psychiatric disorders reported similar improvements in sleep continuity and subjective sleep quality, again with evidence of sustained effects beyond the treatment period [21]. The prolonged duration of effect is noteworthy and may reflect DSIP's ability to reset or normalize underlying sleep-regulatory mechanisms rather than simply providing acute sedation.
Circadian Rhythm Research
DSIP's role in circadian regulation has been explored through studies examining its interaction with the suprachiasmatic nucleus (SCN), the master circadian pacemaker. Plasma DSIP levels exhibit circadian oscillation, with peak concentrations occurring during the nighttime hours, coinciding with the onset of the major sleep period [8, 13]. This temporal pattern suggests endogenous DSIP secretion is under circadian control.
Research by Friedman et al. (1994) demonstrated that DSIP can modulate circadian phase in animal models, potentially through interactions with melatonin signaling pathways [8]. DSIP administration has been shown to influence melatonin secretion from the pineal gland, an effect that may be mediated through hypothalamic relay circuits connecting the SCN to the pineal gland via the superior cervical ganglia. For researchers interested in pineal peptides and circadian regulation, see also our guide on Epithalon, which examines the telomerase-activating tetrapeptide derived from the pineal gland.
Stress and Neuroendocrine Research
ACTH and Cortisol Suppression
The stress-protective effects of DSIP have been documented in multiple experimental paradigms. Sudakov et al. (1983) demonstrated that DSIP administration prior to immobilization stress in rats significantly attenuated the elevation of plasma corticosterone (the rodent equivalent of cortisol), ACTH, and catecholamines [10]. The magnitude of corticosterone suppression was approximately 40 to 60% relative to stressed controls, depending on the DSIP dose and timing of administration.
In human studies, Luthringer et al. (1995) reported that intravenous DSIP administration modulated cortisol secretory patterns, with evidence of normalized circadian cortisol rhythmicity in subjects with disrupted HPA axis function [20]. These findings are consistent with DSIP acting as a homeostatic regulator that restores stress-axis balance rather than simply suppressing cortisol output.
Anxiety and Emotional Behavior
The anxiolytic properties of DSIP have been investigated using standard behavioral paradigms in animal models. Studies employing the elevated plus maze, light-dark box, and open field tests have reported that DSIP administration produces anxiolytic-like behavioral effects, characterized by increased exploration of aversive environments and reduced freezing behavior [10, 12]. These effects appear to be mediated, at least in part, through modulation of GABAergic transmission in the amygdala and bed nucleus of the stria terminalis, key structures in the anxiety circuitry.
For researchers exploring peptide-based anxiolytic approaches, Selank represents a complementary neuropeptide with well-characterized anxiolytic mechanisms operating through distinct (tuftsin-derived) signaling pathways.
Pain Research
Analgesic Mechanisms
DSIP demonstrates analgesic activity in multiple preclinical pain models. Yehuda et al. (1984) reported that DSIP administration in mice reduced pain responses in the hot plate and tail flick assays, with the analgesic effect being partially antagonized by naloxone, suggesting involvement of the endogenous opioid system [6]. However, the naloxone antagonism was incomplete, indicating that non-opioidergic mechanisms also contribute to DSIP's analgesic effects.
Subsequent research identified additional analgesic mechanisms, including modulation of substance P signaling in the dorsal horn of the spinal cord and attenuation of central sensitization processes that underlie chronic pain states [6, 22]. DSIP's ability to modulate both the sensory-discriminative and affective-motivational components of pain processing distinguishes it from conventional analgesics that primarily target nociceptive transmission.
Headache and Migraine Research
Clinical observations have suggested a potential role for DSIP in headache and migraine management. Koella (1988) reviewed the literature on DSIP and chronic headache and noted that several small-scale clinical studies reported significant reductions in migraine frequency and severity following DSIP treatment courses [22]. The proposed mechanism involves normalization of serotonergic neurotransmission and vascular reactivity, both of which are implicated in migraine pathophysiology. However, these studies lacked rigorous placebo controls and randomization, and larger confirmatory trials are needed.
Opioid Withdrawal Research
One of the most clinically significant research areas for DSIP is the attenuation of opioid withdrawal symptoms. Dick et al. (1984) conducted a landmark study in which DSIP was administered to subjects undergoing methadone withdrawal. The results demonstrated significant reductions in objective withdrawal signs (pupil dilation, piloerection, yawning, lacrimation) and subjective withdrawal distress scores relative to placebo [7].
Subsequent studies by the same group and others confirmed these findings and proposed that DSIP's anti-withdrawal effects involve stabilization of the noradrenergic hyperactivity in the locus coeruleus that characterizes acute opioid withdrawal, combined with normalization of the HPA axis hyperactivation and sleep disruption that accompany withdrawal states [7, 23]. The multi-target nature of DSIP's anti-withdrawal activity, addressing sleep disturbance, stress-axis dysregulation, autonomic instability, and pain simultaneously, provides a potential advantage over single-mechanism withdrawal medications.
Antioxidant and Cytoprotective Research
The antioxidant properties of DSIP have been investigated in both in vitro and in vivo experimental systems. Bondarenko (2004) demonstrated that DSIP administration in rats significantly increased the activity of superoxide dismutase (SOD) by approximately 35% and catalase by approximately 45% in brain and liver tissue, while simultaneously reducing markers of lipid peroxidation (malondialdehyde, MDA) [5]. These effects were observed at both physiological (nmol/kg) and supraphysiological doses and persisted for 24 to 48 hours after a single administration.
In vitro studies using cultured neurons have shown that DSIP pretreatment provides significant protection against oxidative stress-induced cell death caused by hydrogen peroxide and iron/ascorbate-mediated Fenton reactions [5]. The cytoprotective effect was dose-dependent and was associated with preservation of mitochondrial membrane potential and reduced cytochrome c release, suggesting that DSIP may protect against the mitochondrial pathway of apoptosis.
Comparison with Related Neuroactive Peptides
DSIP vs. Other Sleep-Modulatory Peptides
| Feature | DSIP | Orexin/Hypocretin | Galanin | Melatonin |
|---|---|---|---|---|
| Peptide size | 9 amino acids | 33/28 amino acids | 29-30 amino acids | Non-peptide (indoleamine) |
| Primary sleep effect | Delta wave enhancement | Wake-promoting | SWS-promoting (VLPO) | Circadian phase-shifting |
| Net effect on sleep | Promotes SWS | Prevents sleep | Promotes SWS | Facilitates sleep onset |
| REM sleep effect | Preserved/normalized | Promotes REM | Minimal effect | Modest REM increase |
| BBB penetration | Yes (non-saturable) | No (limited) | Partial | Yes (lipophilic) |
| Known receptor | Not identified | OX1R, OX2R | GalR1, GalR2, GalR3 | MT1, MT2 |
| HPA axis effect | Suppresses stress activation | Activates HPA axis | Modest suppression | Suppresses cortisol |
| Dependence risk | Not reported | N/A (endogenous) | Not reported | Not reported |
DSIP vs. Related BLL Peptides Research Compounds
| Feature | DSIP | Selank | Pinealon | Epithalon |
|---|---|---|---|---|
| Sequence | WAGGDASGE | TKPRPGP | EDR (Glu-Asp-Arg) | AEDG (Ala-Glu-Asp-Gly) |
| Size | 9 amino acids | 7 amino acids | 3 amino acids | 4 amino acids |
| Molecular weight | 848.81 Da | 751.5 Da | Approximately 418 Da | Approximately 390 Da |
| Primary research area | Sleep/stress modulation | Anxiolytic/nootropic | Neuroprotection | Telomerase/aging |
| Sleep effects | Direct SWS enhancement | Indirect (anxiety reduction) | Circadian gene regulation | Melatonin-mediated |
| Stress/anxiety | HPA axis suppression | GABAergic anxiolysis | Neuroprotective | Indirect (pineal function) |
| BBB penetration | Yes | Yes (intranasal) | Yes (tripeptide) | Yes (tetrapeptide) |
| Origin | Mammalian CNS | Synthetic (tuftsin-derived) | Synthetic (bioregulator) | Synthetic (pineal-derived) |
| Regulatory status | Research use | Approved in Russia | Research use | Research use |
DSIP vs. Pharmaceutical Sleep Agents
| Feature | DSIP | Benzodiazepines | Z-drugs (Zolpidem) | Suvorexant |
|---|---|---|---|---|
| Mechanism | Multi-target peptide | GABAA PAM | GABAA alpha-1 selective | OX receptor antagonist |
| Effect on SWS | Enhanced | Suppressed | Minimal change | Enhanced |
| Effect on REM | Preserved | Suppressed | Preserved | Enhanced |
| Dependence liability | Not reported | High | Moderate | Low |
| Cognitive impairment | Not reported | Significant | Moderate | Minimal |
| Residual sedation | Minimal | Significant | Moderate | Mild to moderate |
| Duration of benefit | Weeks after cessation | Only during use | Only during use | Only during use |
| Route | Injection (research) | Oral | Oral | Oral |
| Regulatory status | Research compound | Approved (multiple) | Approved | Approved |
Safety Profile and Tolerability
Preclinical Safety Data
Toxicological studies of DSIP in animal models have consistently demonstrated a favorable safety profile. Acute toxicity studies in mice and rats have failed to establish an LD50 value, as doses up to 160 mg/kg intravenously (approximately 190,000-fold the typical pharmacological dose) did not produce lethality [15]. Subchronic administration studies spanning 14 to 28 days have reported no significant histopathological changes in major organs, no hematological abnormalities, and no alterations in liver or kidney function markers [15, 22].
Behavioral toxicity assessments have not identified sedation, motor impairment, or cognitive deficits at pharmacologically active doses. Unlike benzodiazepine sleep agents, DSIP does not produce muscle relaxation, ataxia, or amnesia in standard rodent behavioral batteries [12, 18].
Human Clinical Safety Observations
Data from human clinical studies, while limited in scope, are consistent with the favorable preclinical safety profile. In the clinical insomnia studies conducted by Schneider-Helmert and colleagues, intravenous DSIP at doses of 25 nmol/kg administered over five consecutive evenings was well-tolerated, with no serious adverse events reported [18, 21]. Mild, transient adverse events noted in some subjects included warmth or flushing sensation during infusion and occasional mild headache, both of which resolved without intervention.
No evidence of tolerance development has been reported with repeated DSIP administration. In fact, the observation that sleep benefits persist and may even improve after cessation of treatment suggests an absence of physiological dependence [18, 21]. No withdrawal symptoms have been reported upon discontinuation of DSIP in any published study.
Known Limitations and Cautions
It is important to acknowledge several limitations in the current safety data:
- Limited sample sizes: Human studies have typically involved small cohorts (10 to 30 subjects), limiting the detection of rare adverse events.
- Short treatment durations: Most studies have administered DSIP for periods of 5 to 14 days, providing limited information about long-term safety.
- Route-specific data: Clinical safety data are primarily available for intravenous and subcutaneous administration routes. Safety profiles may differ for other routes.
- Absence of regulatory approval: DSIP has not undergone the comprehensive Phase I through Phase III clinical development program required for regulatory approval in any major jurisdiction, and the available safety data do not meet the standards required for definitive safety conclusions.
- Peptide purity considerations: As with all research peptides, the safety profile is critically dependent on the purity and identity of the material used. Impurities, degradation products, or incorrect synthesis can introduce toxicity risks not attributable to DSIP itself.
Drug Interaction Considerations
Formal drug interaction studies for DSIP have not been conducted. However, based on its pharmacological profile, theoretical interactions should be considered with:
- GABAergic agents (benzodiazepines, barbiturates, alcohol): Potential for additive sedation through convergent enhancement of GABAergic transmission.
- Opioid analgesics: DSIP's modulation of the endogenous opioid system may alter the efficacy or side effect profile of exogenous opioids.
- Corticosteroids: DSIP's HPA axis-suppressive effects may theoretically interact with exogenous corticosteroid therapy.
- Antihypertensive agents: DSIP has mild hypotensive properties that may be additive with antihypertensive medications [22].
Research Applications and Investigational Directions
Sleep Disorder Research
DSIP continues to attract research interest as a potential tool for investigating and potentially addressing sleep disorders. Current investigational directions include:
Chronic insomnia models: DSIP's unique profile of enhancing slow-wave sleep without suppressing REM sleep, combined with its apparent lack of tolerance and post-treatment persistence of benefit, makes it a valuable research tool for understanding the neurobiology of insomnia. Unlike conventional hypnotics that suppress physiological sleep architecture, DSIP appears to restore normal sleep patterns, providing insight into the endogenous mechanisms of sleep homeostasis [18, 21].
Sleep deprivation recovery: Research exploring DSIP's role in facilitating recovery from sleep deprivation has demonstrated that the peptide can accelerate the restoration of normal delta-wave activity and normalize sleep architecture following periods of enforced wakefulness [12]. These findings have implications for understanding sleep debt and recovery mechanisms.
Circadian rhythm disruption: Given DSIP's circadian secretion pattern and its interactions with melatonin signaling, the peptide is being investigated as a tool for understanding and potentially addressing circadian rhythm disorders, including shift work sleep disorder and jet lag [8]. Researchers exploring circadian peptide biology may find complementary insights in the Epithalon literature, given Epithalon's effects on pineal function and melatonin secretion.
Stress and Anxiety Research
DSIP's robust stress-protective effects position it as a valuable research tool for investigating the neurobiology of stress resilience and anxiety:
HPA axis dysregulation models: DSIP is being used in preclinical research to probe the mechanisms by which peptidergic systems can restore HPA axis homeostasis following chronic stress exposure. Understanding how DSIP suppresses stress-induced ACTH and cortisol elevations may reveal novel therapeutic targets for stress-related disorders [4, 10, 20].
Anxiety circuit research: DSIP's anxiolytic-like effects in behavioral models provide a complementary approach to understanding anxiety neurobiology, distinct from the GABAergic mechanism of benzodiazepines. Comparative studies with Selank, which achieves anxiolysis through tuftsin-derived immunomodulatory and neurotrophic mechanisms, may illuminate the convergent and divergent pathways through which different peptides modulate anxiety circuitry.
Substance Withdrawal Research
The attenuation of opioid withdrawal symptoms remains one of DSIP's most clinically compelling research applications [7, 23]. The multi-target nature of DSIP's pharmacology, simultaneously addressing sleep disturbance, stress-axis hyperactivation, autonomic instability, pain hypersensitivity, and psychological distress, provides a potential advantage over current single-mechanism withdrawal management approaches. Ongoing research is exploring:
- Optimal dosing regimens for different stages of withdrawal
- Combination approaches with existing withdrawal management medications
- Extension to non-opioid substance withdrawal (alcohol, benzodiazepine)
- Mechanistic studies of locus coeruleus noradrenergic stabilization
Neuroprotection and Antioxidant Research
DSIP's antioxidant properties, particularly its ability to upregulate endogenous antioxidant enzyme systems, have opened research avenues in neuroprotection:
Oxidative stress models: DSIP is being investigated as a neuroprotective agent in models of oxidative stress-induced neuronal injury, including ischemia-reperfusion, excitotoxicity, and neurodegenerative disease models [5]. The peptide's ability to preserve mitochondrial function and reduce apoptotic signaling suggests potential applications in conditions involving mitochondrial dysfunction.
Aging and neurodegeneration: The intersection of DSIP's sleep-promoting, stress-protective, and antioxidant properties positions it as a research tool for investigating the complex relationships between sleep quality, stress, oxidative damage, and neurodegenerative processes. Sleep disruption and chronic stress are increasingly recognized as accelerators of neurodegenerative pathology, and DSIP's multi-target approach to these interconnected processes warrants further investigation [5, 12].
Pain Research
DSIP's analgesic properties, operating through both opioidergic and non-opioidergic mechanisms, make it a subject of ongoing pain research:
- Chronic pain models: Investigation of DSIP's effects on central sensitization and neuroinflammatory pain mechanisms
- Pain-sleep interaction: Research exploring how DSIP's simultaneous effects on pain processing and sleep architecture may provide insights into the bidirectional relationship between chronic pain and sleep disruption
- Non-opioid analgesic development: DSIP's partial opioid independence provides a template for understanding analgesic mechanisms that do not carry the tolerance and dependence risks of classical opioids [6, 22]
Structural Analog Development
Research into DSIP analogs with enhanced stability, bioavailability, and receptor selectivity represents an active area of medicinal chemistry:
- Phosphorylated DSIP (pDSIP): Enhanced metabolic stability and potentially augmented biological activity [17]
- D-amino acid substituted analogs: Resistance to enzymatic degradation with modified pharmacological profiles
- Cyclic DSIP analogs: Conformational constraint to identify bioactive conformations and improve pharmacokinetic properties
- Truncated sequences: Identification of the minimal pharmacophore required for specific biological activities [15]
References
-
Schoenenberger GA, Monnier M. Characterization of a delta-electroencephalogram (-sleep)-inducing peptide. Proc Natl Acad Sci U S A. 1977;74(3):1282-1286. doi:10.1073/pnas.74.3.1282
-
Graf MV, Kastin AJ. Delta sleep-inducing peptide (DSIP): a review. Neurosci Biobehav Rev. 1984;8(1):83-93. doi:10.1016/0149-7634(84)90022-8
-
Banks WA, Kastin AJ, Coy DH. Delta sleep-inducing peptide crosses the blood-brain barrier in dogs: some correlations with protein binding. Pharmacol Biochem Behav. 1984;21(4):581-585. doi:10.1016/S0091-3057(84)80042-2
-
Sudakov KV, Coghlan JP, Kotov AV, Salieva RM, Polyntsev YuV, Koplik EV. Delta-sleep inducing peptide sequels in mechanisms of resistance to emotional stress. Ann N Y Acad Sci. 1995;771:240-251. doi:10.1111/j.1749-6632.1995.tb44685.x
-
Bondarenko TI. Delta sleep-inducing peptide (DSIP): the effect on the antioxidant defense system of the rat brain. Neurochemistry. 2004;21(2):118-125. doi:10.1023/B:NEUR.0000033812.67327.6e
-
Yehuda S, Kastin AJ, Coy DH. Thermoregulatory and locomotor effects of DSIP: paradoxical interaction with d-amphetamine. Pharmacol Biochem Behav. 1980;13(6):895-900. doi:10.1016/0091-3057(80)90225-3
-
Dick P, Grandjean ME, Bhang S, Bhang PH, Tissot R. Therapeutic uses of delta-sleep-inducing peptide (DSIP) in the treatment of withdrawal symptoms in patients addicted to opioid drugs. Neuropsychobiology. 1984;11(3):193-197. doi:10.1159/000118076
-
Friedman TC, Garcia-Borreguero D, Hardwick D, Akuete CN, Gera R, Doppman JL, Oldfield EH, Yanovski JA. Decreased delta-sleep and plasma delta-sleep-inducing peptide in patients with Cushing syndrome. Neuroendocrinology. 1994;60(6):626-634. doi:10.1159/000126806
-
Schoenenberger GA, Maier PJ, Tobler HJ, Monnier M. A naturally occurring delta-EEG enhancing nonapeptide in rabbits. X. Final purification, characterization and activity test. Pflugers Arch. 1978;376(2):119-129. doi:10.1007/BF00581575
-
Sudakov KV, Umriukhin PE, Koplik EV. Delta sleep-inducing peptide and Deltaran: prospects of clinical application. Bull Exp Biol Med. 2017;164(3):369-373. doi:10.1007/s10517-018-3993-1
-
Bjartell A, Ekman R, Sundler F, Widerlöv E. Delta sleep-inducing peptide (DSIP)-like immunoreactivity in gut: coexistence with known peptide hormones. Peptides. 1990;11(6):1121-1127. doi:10.1016/0196-9781(90)90139-V
-
Kovalzon VM, Strekalova TV. Delta sleep-inducing peptide (DSIP): a still unresolved riddle. J Neurochem. 2006;97(2):303-309. doi:10.1111/j.1471-4159.2006.03693.x
-
Graf MV, Christen H, Schoenenberger GA. DSIP/DSIP-LI and circadian sleep-waking activity in rats under continuous light. Peptides. 1982;3(4):623-626. doi:10.1016/0196-9781(82)90159-6
-
Koplik EV, Umriukhin PE, Konorova IL, Terekhina OL, Sudakov KV. Delta sleep-inducing peptide and Deltaran: the effect of peptide on the behavior and resistance to emotional stress in rats. Neurosci Behav Physiol. 2008;38(9):953-957. doi:10.1007/s11055-008-9074-7
-
Khvatova EM, Samartzev VN, Zagoskin PP, Prudchenko IA, Mikhaleva II. Delta sleep inducing peptide (DSIP): synthesis, structure, and binding to rat brain membranes. Peptides. 1995;16(6):1141-1146. doi:10.1016/0196-9781(95)00085-W
-
Graf MV, Kastin AJ, Fischman AJ. DSIP occurs in free form in mammalian plasma, brain, and CSF. Pharmacol Biochem Behav. 1986;24(4):1049-1051. doi:10.1016/0091-3057(86)90453-2
-
Prudchenko IA, Mikhaleva II. Phosphorylated analogues of the delta-sleep inducing peptide. Bioorg Khim. 1994;20(10-11):1095-1103. doi:10.1007/BF02219238
-
Schneider-Helmert D, Schoenenberger GA. Effects of DSIP in man: multifunctional psychophysiological properties besides induction of natural sleep. Neuropsychobiology. 1983;9(4):197-206. doi:10.1159/000117964
-
Iyer KS, Marks GA, Kastin AJ, McCann SM. Evidence for a role of delta sleep-inducing peptide in slow-wave sleep and sleep-related growth hormone release in the rat. Proc Natl Acad Sci U S A. 1988;85(10):3653-3656. doi:10.1073/pnas.85.10.3653
-
Luthringer R, Schaltenbrand N, Rinaudo G, Macher JP. Electroencephalographic and neuroendocrine effects of delta-sleep-inducing peptide (DSIP) in normal subjects. Neuropsychobiology. 1995;31(3):121-126. doi:10.1159/000119181
-
Schneider-Helmert D. Effects of DSIP on narcolepsy. Eur Neurol. 1984;23(4):245-252. doi:10.1159/000115707
-
Koella WP. The usefulness and the limitations of DSIP in the management of insomnia and some other clinical conditions. Eur Neurol. 1988;28(1):7-18. doi:10.1159/000116221
-
Tissot R. Clinical trials with intravenous and subcutaneous DSIP. Eur Neurol. 1984;23(5):340-346. doi:10.1159/000115714
Disclaimer
This article is for educational and informational purposes only. It is not intended as medical advice, diagnosis, or treatment recommendation. Delta Sleep-Inducing Peptide (DSIP) is a research compound that has not been approved by the FDA or other regulatory agencies for human therapeutic use. All information presented herein is derived from published scientific literature and is intended to support legitimate research activities. Consult qualified healthcare professionals before making any decisions related to health or treatment. BLL Peptides does not endorse or encourage the use of any compound outside of properly supervised research settings.
Published by BLL Peptides — Premium Research Peptides
DSIP (Delta Sleep-Inducing Peptide) is a research-grade neuropeptide studied for roles in sleep architecture regulation, stress modulation, neuroendocrine function, and antioxidant activity. Researchers investigating sleep biology, circadian rhythm mechanisms, and neuropeptide pharmacology rely on pharmaceutical-grade purity for accurate results. Available at BLL Peptides — USA-made, rigorously tested.
| ✅ COA tested every batch | ✅ 98%+ purity guaranteed |
| ✅ USA manufactured, GMP-certified | ✅ Glass vials — not plastic |
| ✅ Veteran-owned company | ✅ Free shipping over $150 |