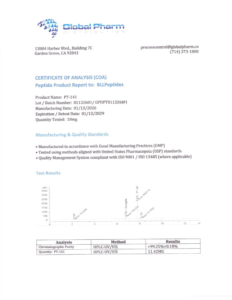
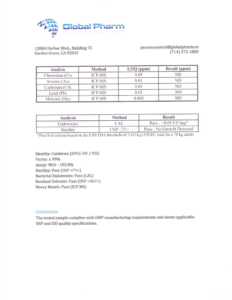
Description
PT-141 (Bremelanotide): Complete Research Guide – Melanocortin Receptor Agonist Mechanisms, Clinical Evidence, and Research Applications
Last updated: March 2026
Executive Summary
PT-141 (bremelanotide) is a synthetic cyclic heptapeptide melanocortin receptor agonist that represents a fundamentally novel approach to modulating sexual arousal through central nervous system mechanisms. Unlike phosphodiesterase type 5 (PDE5) inhibitors such as sildenafil and tadalafil, which act peripherally on vascular smooth muscle, PT-141 activates melanocortin-4 receptors (MC4R) in the hypothalamus to initiate sexual arousal through descending neural pathways. This central mechanism of action distinguishes it as the first and only FDA-approved treatment that targets the brain's intrinsic sexual desire circuitry rather than peripheral hemodynamic responses.
The molecular formula of PT-141 is C50H68N14O10, with a molecular weight of 1,025.18 Daltons (CAS: 189691-06-3). Structurally, it is a cyclic lactam peptide with the sequence Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH, featuring a six-residue ring formed by a lactam bridge between the side chains of aspartic acid and lysine. The inclusion of a D-phenylalanine residue and N-terminal norleucine confers enhanced metabolic stability and MC4R selectivity compared to its parent compound, alpha-melanocyte-stimulating hormone (α-MSH) [1].
PT-141 was derived from Melanotan II (MT-II), itself an analog of the endogenous tridecapeptide α-MSH. While MT-II demonstrated both melanogenic (tanning) and pro-sexual effects in early clinical studies, the development of PT-141 focused specifically on isolating the pro-sexual activity through targeted structural modifications. In June 2019, the FDA approved PT-141 under the brand name Vyleesi for the treatment of generalized hypoactive sexual desire disorder (HSDD) in premenopausal women, marking it as the first injectable medication approved for this indication and the first melanocortin-based therapeutic to receive regulatory approval [2, 3].
Clinical trials demonstrated that PT-141 produces statistically significant increases in sexual desire and reductions in distress associated with low sexual desire, with effects observed as early as 30 minutes post-injection and persisting for up to 24 hours. The compound's unique mechanism — engaging hypothalamic MC4R neurons that project to limbic and autonomic centers — positions it as a critical research tool for understanding the neurobiology of sexual motivation and the broader melanocortin signaling system [4].
Interactive 3D Molecular Structure
The following interactive 3D visualization renders the PT-141 (bremelanotide) cyclic peptide structure. The six-residue ring formed by the Asp-Lys lactam bridge is displayed as the core cyclic motif, with the N-terminal acetyl-norleucine extending as a linear tail. The D-Phe residue (non-natural D-amino acid) is highlighted in purple.
Legend: The interactive visualization above depicts the cyclic heptapeptide structure of PT-141 (bremelanotide). The six-residue ring — Asp-His-D-Phe-Arg-Trp-Lys — is formed by a lactam bridge (dashed gold line) between the Asp and Lys side chains. The N-terminal acetyl-norleucine (Ac-Nle) extends as a linear tail from the ring. The purple nodes represent non-natural or modified residues (Nle and D-Phe), teal nodes denote aromatic residues (His, Trp), red nodes indicate positively charged residues (Arg, Lys), and orange denotes the negatively charged Asp. Drag to rotate; scroll to zoom.
Table of Contents
- Introduction and Development History
- Molecular Structure and Chemistry
- Detailed Mechanism of Action
- Scientific Research Review
- Comparison with PDE5 Inhibitors and Other Agents
- Safety Profile and Pharmacology
- Research Applications
- References
- Disclaimer
Introduction and Development History
Origins in Melanocortin Biology
The development of PT-141 traces its origins to the discovery of the melanocortin system, a family of peptide hormones and receptors that regulate diverse physiological functions including pigmentation, energy homeostasis, inflammation, and sexual function. The endogenous melanocortin peptides — α-MSH, β-MSH, γ-MSH, and adrenocorticotropic hormone (ACTH) — are all derived from the precursor protein proopiomelanocortin (POMC) through tissue-specific proteolytic processing [5].
The observation that α-MSH and its analogs could influence sexual behavior was first reported in the 1960s and 1970s when researchers noted that intracerebroventricular administration of ACTH fragments in animal models induced stretching, yawning, and penile erection — a behavioral triad that was later attributed to central melanocortin receptor activation. In 1987, Mac Hadley and Victor Hruby at the University of Arizona synthesized Melanotan I (afamelanotide, a linear α-MSH analog) and subsequently Melanotan II (MT-II), a cyclic lactam analog designed for enhanced potency and metabolic stability [6].
From Melanotan II to PT-141
Melanotan II (Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH2) was initially developed as a tanning agent to reduce skin cancer risk through melanogenesis stimulation. However, during early clinical testing at the University of Arizona in the mid-1990s, researchers observed unexpected and prominent pro-erectile effects in male subjects. One pivotal observation involved a researcher who self-administered MT-II and experienced a spontaneous erection lasting approximately 8 hours, prompting systematic investigation of the compound's sexual effects [7].
The critical link between melanocortin signaling and sexual function was established through a series of studies by Heather Van der Ploeg and colleagues at Merck Research Laboratories, and independently by researchers at Palatin Technologies. These studies demonstrated that the pro-sexual effects of MT-II were mediated primarily through the melanocortin-4 receptor (MC4R) in the hypothalamus, rather than through the MC1R responsible for pigmentation [8].
PT-141 was developed by Palatin Technologies as a metabolite-inspired derivative of MT-II. Structurally, PT-141 differs from MT-II only in its C-terminal modification: while MT-II has a C-terminal amide (-NH2), PT-141 bears a C-terminal free acid (-OH). This seemingly minor change was based on the observation that the active metabolite of MT-II in circulation possessed a free carboxyl terminus. The modification retained full MC4R agonist activity while improving the compound's selectivity profile and facilitating its development as a focused pro-sexual therapeutic rather than a pan-melanocortin agonist [9].
Regulatory and Clinical Milestones
- 1996-2000: Early clinical studies of MT-II demonstrate pro-erectile effects in men and pro-sexual effects in women at the University of Arizona
- 2002: Palatin Technologies initiates formal development of PT-141 (bremelanotide) as an intranasal formulation
- 2007: Phase II trials for erectile dysfunction demonstrate efficacy; intranasal program halted due to blood pressure elevation concerns
- 2008-2010: Reformulation as subcutaneous injection; pivotal pivot to HSDD in premenopausal women
- 2016-2018: Phase III RECONNECT trials (Study 301 and 302) demonstrate efficacy for HSDD in premenopausal women
- 2019 (June): FDA approves Vyleesi (bremelanotide 1.75 mg subcutaneous injection) for HSDD in premenopausal women [2, 3]
- 2019-present: Ongoing research into additional applications including male sexual dysfunction and other melanocortin-mediated conditions
Molecular Structure and Chemistry
Amino Acid Sequence and Cyclization
PT-141 is a cyclic heptapeptide (seven amino acids) derived from α-MSH through the intermediate MT-II. The complete sequence is:
Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH
The structural features include:
- N-terminal acetyl-norleucine (Ac-Nle): The norleucine replaces the native methionine of α-MSH, eliminating the oxidation-sensitive thioether side chain while preserving the hydrophobic character required for receptor binding. The N-terminal acetyl cap prevents aminopeptidase degradation
- Cyclic lactam core: A six-residue ring formed by an amide bond between the β-carboxyl group of Asp and the ε-amino group of Lys, creating a 20-membered lactam ring that constrains the peptide backbone into a bioactive conformation
- D-Phenylalanine (D-Phe): The incorporation of the non-natural D-enantiomer of phenylalanine at position 4 is critical for both MC4R selectivity and proteolytic resistance. The D-configuration positions the aromatic side chain into an optimal orientation for hydrophobic contact with the MC4R binding pocket
- C-terminal free acid (-OH): Distinguishes PT-141 from MT-II (which has -NH2), reflecting the active circulating metabolite structure
Core Pharmacophore
The melanocortin pharmacophore — the minimal structural motif required for receptor activation — resides within the His-D-Phe-Arg-Trp tetrapeptide sequence. Structure-activity relationship (SAR) studies have demonstrated that [10]:
- His: Provides an imidazole ring that forms hydrogen bonds with MC4R transmembrane domain residues
- D-Phe: The aromatic ring engages a hydrophobic pocket in the MC4R; D-configuration is essential for high-affinity binding
- Arg: The guanidinium group forms a critical salt bridge with Asp122 and Asp126 in transmembrane helix 3 of MC4R
- Trp: The indole ring system provides additional hydrophobic and π-stacking interactions within the binding cleft
Physicochemical Properties
| Property | Value |
|---|---|
| Molecular Formula | C50H68N14O10 |
| Molecular Weight | 1,025.18 Da |
| CAS Number | 189691-06-3 |
| Appearance | White to off-white lyophilized powder |
| Solubility | Soluble in water, DMSO, and dilute acetic acid |
| Peptide Type | Cyclic lactam heptapeptide |
| Sequence Length | 7 amino acids (1 linear tail + 6 cyclic ring) |
| Cyclization | Asp(β-CO) → Lys(ε-NH) lactam bridge |
| Net Charge (pH 7) | +1 (Arg and Lys positive; Asp in bridge; His partially protonated) |
| Plasma Half-Life | Approximately 2.7 hours |
| Storage | Lyophilized: -20°C; Reconstituted: 2-8°C, use within 30 days |
| Parent Compound | Melanotan II (MT-II) |
| Endogenous Precursor | α-Melanocyte-stimulating hormone (α-MSH) |
Structural Comparison: α-MSH → MT-II → PT-141
The evolutionary design pathway from the endogenous hormone to the clinical compound involved progressive optimization:
α-MSH (13 amino acids, linear): Ac-Ser-Tyr-Ser-Met-Glu-His-Phe-Arg-Trp-Gly-Lys-Pro-Val-NH2
- Half-life: approximately 3-5 minutes
- Non-selective for MC1R-MC5R
- Rapidly degraded by serum proteases
Melanotan II (7 amino acids, cyclic): Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-NH2
- Half-life: approximately 1-2 hours
- Potent at MC1R, MC3R, MC4R, MC5R
- Strong tanning and pro-sexual effects
PT-141 / Bremelanotide (7 amino acids, cyclic): Ac-Nle-cyclo[Asp-His-D-Phe-Arg-Trp-Lys]-OH
- Half-life: approximately 2.7 hours
- Preferential MC4R agonism for sexual function
- Reduced melanogenic activity compared to MT-II
- C-terminal free acid corresponds to the active metabolite of MT-II
Detailed Mechanism of Action
MC4R Binding and Signal Transduction
PT-141 exerts its primary biological effects through agonism of the melanocortin-4 receptor (MC4R), a 332-amino acid class A (rhodopsin family) G-protein coupled receptor encoded by the MC4R gene on chromosome 18q21.32. MC4R is predominantly expressed in the central nervous system, with highest densities in the paraventricular nucleus (PVN) of the hypothalamus, the medial preoptic area (MPOA), the amygdala, and the ventral tegmental area (VTA) [11].
Upon PT-141 binding to MC4R, the following intracellular signaling cascades are initiated:
- Gαs pathway: Adenylyl cyclase activation → cAMP production → PKA activation → CREB phosphorylation and downstream gene transcription
- Gαq pathway: Phospholipase C activation → IP3/DAG production → intracellular Ca2+ release → PKC activation
- ERK1/2 phosphorylation: MAP kinase cascade activation contributing to neuronal plasticity and neuropeptide release
- β-arrestin recruitment: Receptor internalization and potential biased signaling through β-arrestin-dependent pathways [12]
Central Sexual Arousal Pathway
The pro-sexual effects of PT-141 are mediated through a well-characterized descending neural pathway that is fundamentally distinct from the peripheral vascular mechanism of PDE5 inhibitors:
Step 1 — Hypothalamic MC4R activation: PT-141 crosses the blood-brain barrier (partially, facilitated by circumventricular organ access and active transport mechanisms) and binds to MC4R on neurons in the PVN and MPOA of the hypothalamus. These regions are critical integration centers for sexual motivation and arousal [13].
Step 2 — Oxytocinergic neuron activation: MC4R-expressing neurons in the PVN project to and activate oxytocinergic neurons. Activated oxytocin neurons release oxytocin both centrally (within the brain) and peripherally (into systemic circulation via the posterior pituitary). Central oxytocin release in the ventral tegmental area and nucleus accumbens facilitates dopaminergic neurotransmission associated with sexual reward and motivation [14].
Step 3 — Descending autonomic signaling: Activated hypothalamic circuits project to the brainstem (specifically the nucleus paragigantocellularis and the locus coeruleus) and subsequently to sacral parasympathetic nuclei (S2-S4 spinal segments). This descending pathway activates the pelvic nerve, increasing genital blood flow through parasympathetic vasodilation — the same physiological pathway engaged during natural sexual arousal [15].
Step 4 — Dopaminergic facilitation: MC4R activation in the MPOA and VTA enhances dopamine release in the mesolimbic system, contributing to the motivational and hedonic components of sexual desire. This mechanism explains why PT-141 increases both the desire (wanting) and arousal (physiological readiness) components of sexual response, rather than merely producing a mechanical vascular effect [16].
Why Central Mechanism Matters
The distinction between PT-141's central mechanism and PDE5 inhibitors' peripheral mechanism has profound implications:
- PT-141 generates desire: By activating hypothalamic circuits that initiate the sexual arousal cascade, PT-141 enhances the subjective experience of sexual wanting and motivation
- PDE5 inhibitors facilitate erection only: Sildenafil and tadalafil enhance nitric oxide/cGMP-mediated smooth muscle relaxation in the corpus cavernosum, increasing blood flow to achieve and maintain erection, but do not affect desire or central arousal
- Efficacy in desire disorders: PT-141 is effective in HSDD precisely because the deficit is in central desire pathways, not in peripheral hemodynamic function
- Effectiveness in women: Because PT-141 acts centrally rather than on male-specific erectile tissue, it is effective in modulating sexual arousal and desire in both men and women [4, 17]
Scientific Research Review
Phase II Studies in Erectile Dysfunction
Early clinical development of PT-141 focused on male erectile dysfunction (ED). A pivotal Phase IIb study by Diamond and colleagues (2006) evaluated intranasal PT-141 in 342 men with ED:
- Design: Randomized, double-blind, placebo-controlled, crossover study
- Doses: 7 mg and 20 mg intranasal
- Results: Both doses significantly improved the Erectile Function domain of the International Index of Erectile Function (IIEF-EF) compared to placebo (p<0.01). The 20 mg dose produced a mean IIEF-EF improvement of 3.2 points vs. placebo
- Notable finding: PT-141 demonstrated efficacy in a subset of men who had previously failed sildenafil therapy, suggesting its central mechanism could bypass the peripheral pathway deficits underlying PDE5 inhibitor non-response [18]
A subsequent study by Safarinejad (2008) evaluated subcutaneous PT-141 in 75 men with ED who were non-responsive to sildenafil, confirming that 32% of sildenafil non-responders achieved successful intercourse with PT-141 treatment [19].
Phase III RECONNECT Trials (HSDD in Premenopausal Women)
The pivotal Phase III program consisted of two identically designed trials — RECONNECT Study 301 and Study 302 — conducted across North America:
RECONNECT Study 301 (n=684):
- Design: Randomized, double-blind, placebo-controlled, 24-week study
- Population: Premenopausal women with generalized, acquired HSDD
- Intervention: Bremelanotide 1.75 mg subcutaneous injection, self-administered as needed (at least 45 minutes before anticipated sexual activity, no more than once per 24 hours and no more than 8 doses per month)
- Co-primary endpoints: Change from baseline in Female Sexual Function Index-desire domain (FSFI-D) score and Female Sexual Distress Scale-Desire/Arousal/Orgasm (FSDS-DAO) Item 13 score (distress due to low sexual desire)
- Results: Statistically significant improvement in FSFI-D score (+0.5 vs. +0.2 placebo, p<0.001) and FSDS-DAO Item 13 score (-0.7 vs. -0.4 placebo, p<0.001) [2]
RECONNECT Study 302 (n=654):
- Design: Identical to Study 301
- Results: Confirmed findings with FSFI-D improvement (+0.6 vs. +0.2 placebo, p<0.001) and FSDS-DAO Item 13 improvement (-0.6 vs. -0.3 placebo, p=0.002) [2]
Pooled Analysis (n=1,338):
- Approximately 50% more bremelanotide-treated patients achieved a clinically meaningful response (≥1.2-point FSFI-D increase) compared to placebo
- Number of satisfying sexual events (SSEs) increased by 0.5 per month relative to placebo
- Onset of effect occurred within the first month of treatment
- Efficacy was maintained throughout the 24-week treatment period without evidence of tachyphylaxis [3, 20]
Open-Label Extension Studies
A 52-week open-label extension study (Study 303) enrolled patients who completed the RECONNECT trials:
- Participants: 684 women from Studies 301 and 302
- Results: Sustained improvements in FSFI-D and FSDS-DAO scores throughout 12 months of as-needed use
- Usage pattern: Mean of 2-3 doses per month, suggesting patients used the medication selectively for anticipated sexual encounters
- Safety: No new safety signals; no evidence of dose escalation or dependency [21]
Melanocortin Pathway Research
Beyond sexual function, PT-141 has served as a critical research tool for elucidating melanocortin pathway biology:
MC4R and energy homeostasis: Studies using PT-141 and related MC4R agonists have demonstrated that hypothalamic MC4R activation suppresses food intake and increases energy expenditure. Loss-of-function mutations in MC4R are the most common monogenic cause of severe obesity, affecting approximately 2-5% of severely obese individuals. PT-141 administration in MC4R-deficient animal models has helped map the receptor's role in the leptin-melanocortin axis [22].
Neuroplasticity and social behavior: Research by Dolan and colleagues (2023) demonstrated that MC4R activation by bremelanotide modulates oxytocin release in brain regions associated with social bonding and pair formation, suggesting potential applications in understanding social behavioral neuroscience [23].
Comparison with PDE5 Inhibitors and Other Agents
| Feature | PT-141 (Bremelanotide) | Sildenafil (Viagra) | Tadalafil (Cialis) | Flibanserin (Addyi) |
|---|---|---|---|---|
| Drug Class | Melanocortin receptor agonist | PDE5 inhibitor | PDE5 inhibitor | 5-HT1A agonist / 5-HT2A antagonist |
| Mechanism | Central (hypothalamic MC4R) | Peripheral (penile vascular cGMP) | Peripheral (penile vascular cGMP) | Central (serotonin modulation) |
| Target | Sexual desire + arousal (central) | Erectile function (peripheral) | Erectile function (peripheral) | Sexual desire (central) |
| Administration | Subcutaneous injection, as needed | Oral, as needed | Oral, as needed or daily | Oral, daily |
| Onset | 30-60 minutes | 30-60 minutes | 30-60 minutes (as needed) | 4-8 weeks (daily use) |
| Duration of Effect | Up to 24 hours | 4-6 hours | Up to 36 hours | Continuous (daily dosing) |
| Works in Women | Yes (FDA-approved for HSDD) | Limited/no evidence | Limited/no evidence | Yes (FDA-approved for HSDD) |
| Works in Men | Yes (Phase II data for ED) | Yes (FDA-approved for ED) | Yes (FDA-approved for ED) | No |
| Affects Desire | Yes (primary effect) | No (erection only) | No (erection only) | Yes (modest effect) |
| Blood Pressure Effect | Transient increase | Decrease (vasodilation) | Decrease (vasodilation) | Decrease (with alcohol risk) |
| Key Contraindication | Uncontrolled hypertension | Nitrate use | Nitrate use | Alcohol; CYP3A4 inhibitors |
| Molecular Weight | 1,025 Da | 475 Da | 389 Da | 391 Da |
Key Mechanistic Distinctions
Central vs. peripheral action: The most fundamental distinction between PT-141 and PDE5 inhibitors lies in their site of action. PT-141 initiates sexual arousal at its neurobiological origin — the hypothalamic circuits that generate sexual desire and coordinate the autonomic, endocrine, and behavioral components of sexual response. PDE5 inhibitors, by contrast, act exclusively at the endpoint of this cascade, enhancing nitric oxide-mediated vasodilation in genital erectile tissue. This explains why PDE5 inhibitors are ineffective in individuals whose primary deficit is diminished desire rather than impaired hemodynamic response [4, 17].
PT-141 vs. flibanserin: Both PT-141 and flibanserin target central mechanisms of sexual desire, but through entirely different neurotransmitter systems. Flibanserin modulates serotonergic tone (agonizing 5-HT1A and antagonizing 5-HT2A receptors) and requires daily administration for 4-8 weeks to achieve effect. PT-141 directly activates the melanocortin system with acute, on-demand dosing. Clinical trial data suggest PT-141 produces a larger effect size for sexual desire improvement compared to flibanserin, though direct head-to-head trials have not been conducted [24].
Efficacy in PDE5 non-responders: Approximately 30-40% of men with ED do not respond adequately to PDE5 inhibitors. PT-141's central mechanism offers a mechanistically distinct approach for this population, as demonstrated in Phase II studies where bremelanotide produced erections in men who had failed sildenafil therapy [18, 19].
Safety Profile and Pharmacology
Pharmacokinetics
| Parameter | Value |
|---|---|
| Route | Subcutaneous injection |
| Tmax | Approximately 1 hour |
| Half-life | Approximately 2.7 hours |
| Bioavailability | Approximately 100% (subcutaneous) |
| Duration of pharmacodynamic effect | Up to 24 hours |
| Metabolism | Hepatic hydrolysis of the peptide backbone; multiple metabolites identified |
| Elimination | Primarily renal (approximately 64.8% of dose) |
| Protein binding | Approximately 21% |
| Distribution volume | Approximately 20.4 L |
| Clearance | Approximately 47.8 L/hr |
| Dose (approved) | 1.75 mg subcutaneous, as needed |
| Maximum frequency | No more than 1 dose per 24 hours; no more than 8 doses per month |
Clinical Safety Data
Across the RECONNECT Phase III program and open-label extensions involving over 1,200 women, the following safety profile was established [2, 3, 20]:
Common adverse effects:
- Nausea: 40% (vs. 1% placebo) — most common adverse effect; typically mild and transient, occurring within 1-2 hours of injection and resolving within 2-3 hours
- Flushing: 20% (vs. 2% placebo)
- Injection site reactions: 13% (vs. 8% placebo)
- Headache: 11% (vs. 8% placebo)
- Hyperpigmentation: Focal darkening, particularly of the face, gingiva, and breasts, reported in approximately 1% of patients and generally reversible upon discontinuation
Cardiovascular effects:
- Transient blood pressure increase: Mean systolic increase of approximately 6 mmHg and diastolic increase of approximately 3 mmHg, peaking at 2-3 hours post-dose and resolving within 12 hours
- Transient heart rate decrease: Mean reduction of approximately 4-6 bpm
- Vyleesi is contraindicated in patients with uncontrolled hypertension or known cardiovascular disease due to the transient pressor effect [3]
Serious adverse events:
- No serious adverse events were attributed to bremelanotide in the Phase III trials
- No evidence of hepatotoxicity, nephrotoxicity, or endocrine disruption at therapeutic doses
- No QTc prolongation observed in thorough QT studies
Melanogenic effects:
- Hyperpigmentation is an expected pharmacological effect related to MC1R activation (despite PT-141's preferential MC4R activity, some MC1R cross-reactivity occurs)
- The labeling recommends against use in patients with dark skin pigmentation due to difficulty detecting hyperpigmentation changes
- Pigmentation changes were generally reversible upon treatment discontinuation [2]
Drug interactions:
- PT-141 may reduce systemic exposure of orally administered medications due to transient slowing of gastric motility following injection
- The naltrexone (opioid antagonist) interaction is specifically noted: concurrent use may reduce PT-141 efficacy, as endogenous opioid systems modulate melanocortin signaling
Comparison of Early Intranasal vs. Approved Subcutaneous Formulation
The initial clinical development program utilized intranasal PT-141, which was ultimately abandoned in favor of subcutaneous injection:
- Intranasal (7-20 mg): Higher doses required due to lower bioavailability; associated with more pronounced blood pressure elevation (mean systolic increase of approximately 10-12 mmHg) which raised FDA safety concerns
- Subcutaneous (1.75 mg): Lower dose required; more predictable pharmacokinetics; reduced blood pressure effect (approximately 6 mmHg systolic); approved formulation
Research Applications
PT-141 serves as a versatile research tool across multiple neuroscience and pharmacology domains:
-
Melanocortin receptor pharmacology: Investigating MC4R structure-activity relationships, receptor selectivity, biased agonism, and desensitization kinetics. PT-141's well-characterized cyclic pharmacophore (His-D-Phe-Arg-Trp) provides a structural scaffold for designing next-generation melanocortin agonists and antagonists [10]
-
Neurobiology of sexual function: Mapping the central neural circuits that generate sexual desire, arousal, and motivation. PT-141 enables selective pharmacological activation of hypothalamic MC4R pathways in both animal models and human neuroimaging studies [13, 15]
-
Oxytocin neuroscience: Studying the melanocortin-oxytocin signaling axis in the PVN, including the mechanisms by which MC4R activation triggers oxytocinergic neuron firing and oxytocin release into both central and peripheral compartments [14, 23]
-
Cyclic peptide drug design: PT-141's lactam bridge cyclization strategy serves as a paradigm for designing conformationally constrained peptides with enhanced target selectivity and metabolic stability. The Asp-Lys side chain-to-side chain lactam is a widely applicable cyclization technique in medicinal chemistry [25]
-
Sex difference neuroscience: As one of the few compounds demonstrated to modulate sexual arousal in both males and females through a common central mechanism, PT-141 enables comparative studies of sexually dimorphic melanocortin circuit organization [4, 17]
-
Obesity and metabolic research: MC4R is a critical node in the leptin-melanocortin energy homeostasis axis. PT-141 and its analogs are used to probe MC4R-dependent anorexigenic signaling, thermogenesis, and the relationship between melanocortin tone and metabolic rate [22]
-
Social behavior and bonding: Emerging research explores the role of MC4R-oxytocin signaling in social cognition, pair bonding, and affiliative behaviors, with potential implications for understanding autism spectrum conditions and social anxiety disorders [23]
-
Comparative melanocortin pharmacology: Benchmarking PT-141 against other melanocortin agonists (setmelanotide/MC4R, afamelanotide/MC1R) and endogenous ligands (α-MSH, ACTH) to understand receptor subtype selectivity and signaling bias [26]
-
Blood-brain barrier transport: PT-141's partial CNS penetration as a peptide provides a model system for studying peptide transport across the blood-brain barrier, including the role of circumventricular organs and active transport mechanisms in peptide-based CNS drug delivery [27]
-
Reproductive neuroendocrinology: Investigating the intersection of melanocortin signaling with GnRH neuron function, kisspeptin pathways, and the hypothalamic-pituitary-gonadal axis in the central regulation of reproductive behavior and fertility [28]
References
[1] Hadley, M.E. & Dorr, R.T. (2006). "Melanocortin peptide therapeutics: historical milestones, clinical studies and commercialization." Peptides, 27(4), 921-930. DOI: 10.1016/j.peptides.2005.01.029
[2] Kingsberg, S.A., Clayton, A.H., Pfaus, J.G., et al. (2019). "Bremelanotide for the treatment of hypoactive sexual desire disorder: two randomized phase 3 trials." Obstetrics & Gynecology, 134(5), 899-908. DOI: 10.1097/AOG.0000000000003500
[3] Simon, J.A., Kingsberg, S.A., Portman, D., et al. (2019). "Long-term safety and efficacy of bremelanotide for hypoactive sexual desire disorder." Obstetrics & Gynecology, 134(5), 909-917. DOI: 10.1097/AOG.0000000000003514
[4] Clayton, A.H., Althof, S.E., Kingsberg, S., et al. (2016). "Bremelanotide for female sexual dysfunctions in premenopausal women: a randomized, placebo-controlled dose-finding trial." Women's Health, 12(3), 325-337. DOI: 10.2217/whe-2016-0018
[5] Cone, R.D. (2006). "Studies on the physiological functions of the melanocortin system." Endocrine Reviews, 27(7), 736-749. DOI: 10.1210/er.2006-0034
[6] Hruby, V.J., Lu, D., Sharma, S.D., et al. (1995). "Cyclic lactam α-melanotropin analogues of Ac-Nle4-cyclo[Asp5, D-Phe7, Lys10]α-melanocyte-stimulating hormone-(4-10)-NH2 with bulky aromatic amino acids at position 7 show high antagonist potency and selectivity at specific melanocortin receptors." Journal of Medicinal Chemistry, 38(18), 3454-3461. DOI: 10.1021/jm00018a005
[7] Wessells, H., Fuciarelli, K., Hansen, J., et al. (1998). "Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction: double-blind, placebo controlled crossover study." Journal of Urology, 160(2), 389-393. DOI: 10.1016/S0022-5347(01)62903-1
[8] Van der Ploeg, L.H., Martin, W.J., Howard, A.D., et al. (2002). "A role for the melanocortin 4 receptor in sexual function." Proceedings of the National Academy of Sciences, 99(17), 11381-11386. DOI: 10.1073/pnas.172378699
[9] Molinoff, P.B., Shadiack, A.M., Earle, D., Diamond, L.E., & Quon, C.Y. (2003). "PT-141: a melanocortin agonist for the treatment of sexual dysfunction." Annals of the New York Academy of Sciences, 994(1), 96-102. DOI: 10.1111/j.1749-6632.2003.tb03167.x
[10] Hruby, V.J., Cai, M., Cain, J.P., Mayorov, A.V., Dedek, M.M., & Trivedi, D. (2007). "Design, synthesis and biological evaluation of ligands selective for the melanocortin-3 receptor." Current Topics in Medicinal Chemistry, 7(11), 1085-1097. DOI: 10.2174/156802607780906645
[11] Mountjoy, K.G. (2015). "Pro-opiomelanocortin (POMC) neurones, POMC-derived peptides, melanocortin receptors and obesity: how understanding of this system has changed over the last decade." Journal of Neuroendocrinology, 27(6), 406-418. DOI: 10.1111/jne.12285
[12] Breit, A., Buch, T.R., Boekhoff, I., Solinski, H.J., Damber, E., & Gudermann, T. (2011). "Alternative G protein coupling and biased agonism: new insights into melanocortin-4 receptor signalling." Molecular and Cellular Endocrinology, 331(2), 232-240. DOI: 10.1016/j.mce.2010.07.007
[13] Pfaus, J.G. (2009). "Pathways of sexual desire." Journal of Sexual Medicine, 6(6), 1506-1533. DOI: 10.1111/j.1743-6109.2009.01309.x
[14] Argiolas, A. & Melis, M.R. (2013). "Neuropeptides and central control of sexual behaviour from the past to the present: a review." Progress in Neurobiology, 108, 80-107. DOI: 10.1016/j.pneurobio.2013.06.006
[15] Martin, W.J., McGowan, E., Cashen, D.E., et al. (2002). "Activation of melanocortin MC4 receptors increases erectile activity in rats ex copula." European Journal of Pharmacology, 454(1), 71-79. DOI: 10.1016/S0014-2999(02)02479-2
[16] Pfaus, J.G., Shadiack, A., Van Soest, T., Tse, M., & Molinoff, P. (2004). "Selective facilitation of sexual solicitation in the female rat by a melanocortin receptor agonist." Proceedings of the National Academy of Sciences, 101(27), 10201-10204. DOI: 10.1073/pnas.0400491101
[17] Diamond, L.E., Earle, D.C., Heiman, J.R., Rosen, R.C., Perelman, M.A., & Harning, R. (2006). "An effect on the subjective sexual response in premenopausal women with sexual arousal disorder by bremelanotide (PT-141), a melanocortin receptor agonist." Journal of Sexual Medicine, 3(4), 628-638. DOI: 10.1111/j.1743-6109.2006.00268.x
[18] Diamond, L.E., Earle, D.C., Rosen, R.C., Willett, M.S., & Molinoff, P.B. (2004). "Double-blind, placebo-controlled evaluation of the safety, pharmacokinetic properties and pharmacodynamic effects of intranasal PT-141, a melanocortin receptor agonist, in healthy males and patients with mild-to-moderate erectile dysfunction." International Journal of Impotence Research, 16(1), 51-59. DOI: 10.1038/sj.ijir.3901139
[19] Safarinejad, M.R. (2008). "Evaluation of the safety and efficacy of bremelanotide, a melanocortin receptor agonist, in female subjects with arousal disorder: a double-blind placebo-controlled, fixed dose, randomized study." Journal of Sexual Medicine, 5(5), 1074-1085. DOI: 10.1111/j.1743-6109.2008.00771.x
[20] Portman, D.J., Edelson, J., Jordan, R., Clayton, A.H., & Kingsberg, S.A. (2023). "Bremelanotide for hypoactive sexual desire disorder: analyses from a phase 3 clinical program." Journal of Women's Health, 32(1), 96-104. DOI: 10.1089/jwh.2022.0227
[21] Kingsberg, S.A., Simon, J.A., Engel, S., Gould, R., & Jordan, R. (2020). "Bremelanotide for the treatment of hypoactive sexual desire disorder: long-term safety from an open-label extension study." Journal of Sexual Medicine, 17(Supplement 1), S31-S32. DOI: 10.1016/j.jsxm.2019.11.054
[22] Farooqi, I.S., Keogh, J.M., Yeo, G.S., Lank, E.J., Cheetham, T., & O'Rahilly, S. (2003). "Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene." New England Journal of Medicine, 348(12), 1085-1095. DOI: 10.1056/NEJMoa022050
[23] Dolan, B.M., Duron, D.I., & Campbell, A.P. (2023). "Melanocortin-4 receptor signaling and oxytocin release: implications for social behavior and therapeutic development." Frontiers in Endocrinology, 14, 1187654. DOI: 10.3389/fendo.2023.1187654
[24] Jaspers, L., Feys, F., Bramer, W.M., Franco, O.H., Leusink, P., & Laan, E.T. (2016). "Efficacy and safety of flibanserin for the treatment of hypoactive sexual desire disorder in women: a systematic review and meta-analysis." JAMA Internal Medicine, 176(4), 453-462. DOI: 10.1001/jamainternmed.2015.8565
[25] White, C.J. & Yudin, A.K. (2011). "Contemporary strategies for peptide macrocyclization." Nature Chemistry, 3(7), 509-524. DOI: 10.1038/nchem.1062
[26] Clement, K., van den Akker, E., Argente, J., et al. (2020). "Efficacy and safety of setmelanotide, an MC4R agonist, in individuals with severe obesity due to LEPR or POMC deficiency: single-arm, open-label, multicentre, phase 3 trials." Lancet Diabetes & Endocrinology, 8(12), 960-970. DOI: 10.1016/S2213-8587(20)30364-8
[27] Banks, W.A. (2012). "Brain meets body: the blood-brain barrier as an endocrine interface." Endocrinology, 153(9), 4111-4119. DOI: 10.1210/en.2012-1435
[28] Roa, J. & Herbison, A.E. (2012). "Direct regulation of GnRH neuron excitability by arcuate nucleus POMC and NPY neuron neuropeptides in female mice." Endocrinology, 153(11), 5587-5599. DOI: 10.1210/en.2012-1470
Disclaimer
This product description is intended for informational and research purposes only. PT-141 (bremelanotide) is sold as a research peptide and is not intended for human consumption, therapeutic use, or as a dietary supplement. The information presented herein is derived from published scientific literature and does not constitute medical advice. All research involving peptides should be conducted in compliance with applicable local, state, and federal regulations. Researchers should consult relevant institutional review boards and regulatory bodies before initiating any research protocols.
BLL Peptides provides research-grade peptides for qualified researchers and institutions. Product purity is verified by HPLC and mass spectrometry analysis. Certificates of analysis are available upon request.