GHRP-6 10mg (3ml)
GHRP-6 is a research-grade growth hormone releasing hexapeptide studied for GH secretion stimulation, appetite promotion, and tissue repair support through GH-dependent metabolic pathways. Researchers investigating the growth hormone axis and body composition rely on pharmaceutical-grade purity for valid experimental outcomes. Available at BLL Peptides — USA-made, rigorously tested. ✅ COA tested every batch✅ 98%+ purity…
Description
GHRP-6: Complete Research Guide – Growth Hormone Releasing Peptide-6 Mechanisms, Ghrelin Mimetic Research, and Endocrine Applications
Last updated: March 2026
Executive Summary
Growth Hormone Releasing Peptide-6 (GHRP-6) is a synthetic hexapeptide growth hormone secretagogue (GHS) with the amino acid sequence His-D-Trp-Ala-Trp-D-Phe-Lys-NH2. Developed by Cyril Y. Bowers and Frank Momany at Tulane University in the early 1980s, GHRP-6 holds a singular place in endocrine pharmacology as the original growth hormone-releasing peptide — the compound whose discovery launched an entirely new field of neuroendocrine research and ultimately led to the identification of the growth hormone secretagogue receptor (GHS-R1a) and the endogenous hormone ghrelin [1].
GHRP-6 acts as a potent agonist of the GHS-R1a receptor, stimulating growth hormone (GH) release from anterior pituitary somatotrophs through Gq/11-mediated phospholipase C activation and intracellular calcium mobilization. Its molecular formula is C46H56N12O6, with a molecular weight of approximately 873.01 Daltons (CAS: 87616-84-0). As a first-generation GHS, GHRP-6 exhibits a broad endocrine activation profile: in addition to robust GH release, it produces significant appetite stimulation through ghrelin-mimetic activity on hypothalamic orexigenic circuits, and elevates both cortisol and prolactin at GH-effective doses [2]. These properties distinguish GHRP-6 from later, more selective secretagogues such as ipamorelin, which was specifically engineered to eliminate the appetite-stimulating and adrenocorticotropic effects characteristic of the original GHRP series.
The historical importance of GHRP-6 cannot be overstated. Its existence as a synthetic ligand for an unknown receptor drove the identification and cloning of GHS-R1a by Howard and colleagues in 1996 [3], which in turn enabled the reverse pharmacology approach that led Kojima, Kangawa, and colleagues to discover ghrelin — the endogenous 28-amino acid acylated peptide hormone — in 1999 [4]. GHRP-6 thus represents one of the most productive examples of a synthetic compound guiding the discovery of an entirely new endogenous signaling system. Beyond its role in GH axis research, GHRP-6 has demonstrated significant gastroprotective, cardioprotective, and anti-inflammatory properties in preclinical models, expanding its research relevance into domains well beyond classical neuroendocrinology [5, 6].
Interactive 3D Molecular Structure
The following interactive 3D visualization renders the GHRP-6 hexapeptide in a ball-and-stick representation. Because GHRP-6 contains only six residues, each amino acid is displayed as a large sphere with connecting bonds, clearly illustrating the two D-amino acid modifications (D-Trp2, D-Phe5) that confer proteolytic resistance and receptor binding specificity, as well as the paired aromatic tryptophan residues (D-Trp2 and Trp4) that are critical for GHS-R1a engagement.
Legend: The interactive visualization above depicts GHRP-6's six-residue structure in a ball-and-stick representation. The two D-amino acid residues — D-Trp (D-tryptophan) and D-Phe (D-phenylalanine) — are highlighted in purple, reflecting their critical role in conferring receptor affinity and proteolytic resistance. Histidine and tryptophan are shown in cyan (aromatic), alanine in gray-blue (hydrophobic), lysine in red (positively charged), and the C-terminal amide (NH2) in gold. Drag to rotate the structure; scroll to zoom.
Table of Contents
- Introduction and Development History
- Molecular Structure and Chemistry
- Detailed Mechanism of Action
- Scientific Research Review
- Comparison with Other GH Secretagogues
- Pharmacokinetics and Safety Profile
- Research Applications
- References
- Disclaimer
Introduction and Development History
The Origin of Growth Hormone-Releasing Peptides
The discovery of growth hormone-releasing peptides traces to the seminal observations of Cyril Y. Bowers at Tulane University in the late 1970s. Bowers, working in collaboration with Frank Momany at the University of Texas, observed that certain synthetic analogs of Met-enkephalin — small opioid peptides — could stimulate the release of growth hormone from anterior pituitary cells in vitro through a mechanism that was completely independent of the growth hormone-releasing hormone (GHRH) receptor [1]. This was a landmark observation, because at the time, GHRH (discovered in 1982 by Guillemin and Rivier) was believed to be the sole hypothalamic factor capable of stimulating GH secretion. The existence of a second, independent GH-releasing pathway was entirely unexpected and initiated a new chapter in neuroendocrine pharmacology.
The initial enkephalin analogs studied by Bowers possessed only weak GH-releasing activity. However, through systematic structure-activity relationship (SAR) studies conducted throughout the early 1980s, Bowers and Momany progressively optimized the peptide scaffold, removing opioid receptor activity while enhancing GH-releasing potency. This iterative chemical evolution culminated in 1984 with the synthesis and characterization of GHRP-6 — the first peptide to demonstrate potent, specific, and reproducible GH-releasing activity both in vitro and in vivo [1]. GHRP-6 thus became the founding member of an entirely new class of endocrine pharmacological agents.
GHRP-6: The Founding Compound
GHRP-6 (His-D-Trp-Ala-Trp-D-Phe-Lys-NH2) was designated as the sixth in a series of growth hormone-releasing peptide candidates evaluated during the Bowers-Momany optimization program. Its hexapeptide structure incorporates two D-amino acid residues (D-Trp at position 2 and D-Phe at position 5) that were found to be essential for both GH-releasing activity and metabolic stability. The inclusion of D-amino acids was a deliberate design strategy: these non-natural stereoisomers resist cleavage by endogenous peptidases that preferentially recognize L-amino acid substrates, thereby extending the peptide's biological half-life [7].
Early studies by Bowers and colleagues demonstrated that GHRP-6 produced dose-dependent GH release from rat anterior pituitary cell cultures, with an efficacy comparable to GHRH but operating through a mechanistically distinct pathway [1]. Crucially, GHRP-6-stimulated GH release was not blocked by GHRH receptor antagonists and did not involve cAMP signaling, confirming the existence of a novel receptor and signaling mechanism. In vivo administration of GHRP-6 to rats, dogs, and humans produced rapid, robust elevations in circulating GH levels, establishing the compound's translational pharmacology across species [8].
From GHRP-6 to the Discovery of GHS-R1a
The most profound scientific contribution of GHRP-6 was its role as the pharmacological tool that drove the identification of its own receptor. For over a decade following GHRP-6's characterization, the receptor mediating its GH-releasing effects remained unknown. Researchers knew that the receptor existed — GHRP-6 binding sites could be identified in pituitary membranes — but molecular cloning efforts proved difficult because no endogenous ligand was available to guide the search.
The breakthrough came in 1996, when Howard and colleagues at Merck Research Laboratories used the non-peptidyl GH secretagogue MK-0677 (which was itself derived from the GHRP pharmacophore) as a probe ligand in an expression cloning strategy. They identified and cloned the growth hormone secretagogue receptor type 1a (GHS-R1a), a seven-transmembrane G protein-coupled receptor (GPCR) expressed predominantly in the pituitary and hypothalamus [3]. This receptor was subsequently shown to bind all known GHRPs, including GHRP-6, with high affinity. The cloning of GHS-R1a was a direct consequence of GHRP-6's existence as a pharmacological tool.
The Road to Ghrelin: Reverse Pharmacology in Action
With GHS-R1a cloned but its endogenous ligand still unknown, the receptor became an "orphan" GPCR — a cloned receptor waiting for its natural agonist. In 1999, Kojima, Hosoda, and Kangawa at the National Cardiovascular Center Research Institute in Osaka employed a reverse pharmacology approach, screening tissue extracts for the ability to activate GHS-R1a in a cell-based calcium mobilization assay. They identified a novel 28-amino acid peptide from rat stomach extracts that potently activated GHS-R1a. This peptide, which they named ghrelin (from the Proto-Indo-European root "ghre," meaning to grow), was found to require an unusual octanoyl modification on its Ser3 residue for receptor activation [4].
The discovery of ghrelin was a watershed moment in endocrinology, revealing a new gut-brain hormonal axis with far-reaching implications for appetite regulation, energy homeostasis, glucose metabolism, and cardiovascular function. Yet this entire discovery cascade — from GHRP-6 to GHS-R1a to ghrelin — originated with Bowers' observation that synthetic enkephalin analogs could release growth hormone. GHRP-6 thus stands as one of the most consequential synthetic peptides in the history of endocrine research, a compound whose creation opened the door to an entirely new hormonal system [2].
Subsequent GHRP Development
Following GHRP-6's success, Bowers and Momany continued SAR optimization of the hexapeptide scaffold. This work produced GHRP-2 (D-Ala-D-2-Nal-Ala-Trp-D-Phe-Lys-NH2), which incorporated a D-2-naphthylalanine residue at position 2 and achieved approximately 2-3 fold greater GH-releasing potency than GHRP-6, along with a somewhat more favorable cortisol/prolactin ratio [9]. Hexarelin (His-D-2-Me-Trp-Ala-Trp-D-Phe-Lys-NH2) represented another GHRP-6 analog featuring a methylated tryptophan at position 2. Later, the pentapeptide ipamorelin (Aib-His-D-2-Nal-D-Phe-Lys-NH2), developed by Novo Nordisk, achieved remarkable GH selectivity by eliminating the cortisol, prolactin, and appetite-stimulating effects characteristic of GHRP-6 and GHRP-2 [10]. Each successive compound in this lineage owed its conceptual and structural foundation to GHRP-6.
Nomenclature and Classification
GHRP-6 is classified within the growth hormone secretagogue (GHS) family as a first-generation peptidyl ghrelin receptor (GHS-R1a) agonist. Its established nomenclature includes:
- Common name: GHRP-6, Growth Hormone Releasing Peptide-6
- Chemical classification: Synthetic hexapeptide growth hormone secretagogue
- CAS Number: 87616-84-0
- Sequence: His-D-Trp-Ala-Trp-D-Phe-Lys-NH2
- Receptor target: GHS-R1a (ghrelin receptor)
The compound should be distinguished from its structural analogs GHRP-2 (D-Ala-D-2-Nal-Ala-Trp-D-Phe-Lys-NH2), hexarelin (His-D-2-Me-Trp-Ala-Trp-D-Phe-Lys-NH2), and ipamorelin (Aib-His-D-2-Nal-D-Phe-Lys-NH2), as well as the non-peptidyl GHS MK-677 (ibutamoren).
Molecular Structure and Chemistry
Amino Acid Sequence
GHRP-6 is a linear hexapeptide with the following primary sequence:
His-D-Trp-Ala-Trp-D-Phe-Lys-NH2
This six-residue peptide incorporates two D-amino acid modifications and a C-terminal amidation, each serving specific pharmacological functions:
-
His (L-histidine, position 1): The N-terminal histidine provides an imidazole ring capable of hydrogen bonding and protonation-dependent charge interactions. At physiological pH, the imidazole side chain (pKa approximately 6.0) exists in a partial protonation equilibrium, enabling pH-sensitive interactions with receptor residues. Histidine at position 1 is unique to GHRP-6 among the major GHRPs; in GHRP-2, this position is occupied by D-alanine, and the replacement of histidine with a non-aromatic D-amino acid was one of the key modifications that enhanced GHRP-2's potency [7].
-
D-Trp (D-tryptophan, position 2): The D-enantiomer of tryptophan at position 2 is a defining feature of the GHRP-6 structure. The indole ring of D-tryptophan provides a large aromatic surface for hydrophobic stacking interactions with the GHS-R1a transmembrane binding pocket. The D-configuration confers resistance to endopeptidases and chymotrypsin-like proteases that preferentially cleave peptide bonds adjacent to L-aromatic residues. Structure-activity studies demonstrated that substitution of D-Trp2 with L-Trp abolished GH-releasing activity, confirming that the D-configuration is essential for correct pharmacophoric geometry at this position [7, 11].
-
Ala (L-alanine, position 3): A small, hydrophobic amino acid that serves as a structural spacer between the aromatic residues at positions 2 and 4. Alanine's minimal side chain (methyl group) avoids steric clashes within the receptor binding site while maintaining appropriate inter-residue spacing for the pharmacophore. This position is conserved across GHRP-6, GHRP-2, and hexarelin [7].
-
Trp (L-tryptophan, position 4): The L-tryptophan at position 4 is highly conserved across the GHRP family and is considered essential for GHS-R1a activation. Its indole ring engages in aromatic stacking interactions with Phe279 and Phe312 within the GHS-R1a transmembrane domain, and removal or substitution of this residue dramatically reduces GH-releasing potency [11, 12]. The pairing of two tryptophan residues (D-Trp2 and L-Trp4) in the GHRP-6 sequence provides a bis-indole aromatic framework that is a signature structural feature of this peptide.
-
D-Phe (D-phenylalanine, position 5): Conserved across all GHRP family members, D-phenylalanine at position 5 is critical for GHS-R1a binding. SAR studies demonstrated that substitution with L-Phe or deletion of this residue eliminates GH-releasing activity entirely, establishing D-Phe5 as an indispensable pharmacophoric element [7]. The phenyl ring contributes hydrophobic contacts within the receptor binding cavity, and the D-configuration ensures both proteolytic resistance and optimal three-dimensional presentation.
-
Lys-NH2 (L-lysine amide, position 6): The C-terminal lysine residue carries a positively charged epsilon-amino group (pKa approximately 10.5) at physiological pH, enabling ionic interactions with negatively charged receptor residues. C-terminal amidation neutralizes the carboxylate negative charge that would otherwise be present at the peptide's C-terminus, improving membrane permeability and enhancing receptor binding through additional hydrogen bond donation capacity. Removal of the amide group reduces GH-releasing potency by approximately 10-fold [1].
Physicochemical Properties
| Property | Value |
|---|---|
| Molecular Formula | C46H56N12O6 |
| Molecular Weight | Approximately 873.01 Da |
| CAS Number | 87616-84-0 |
| Sequence | His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 |
| Sequence Length | 6 amino acids (hexapeptide) |
| D-amino Acid Positions | Position 2 (D-Trp), Position 5 (D-Phe) |
| Appearance | White to off-white lyophilized powder |
| Solubility | Freely soluble in water, DMSO; soluble in dilute acetic acid |
| pI (Isoelectric Point) | Approximately 8.8 |
| Net Charge at pH 7.4 | +2 (from His imidazolium and Lys epsilon-amino group) |
| Storage | -20C lyophilized; 2-8C reconstituted (use within 21 days) |
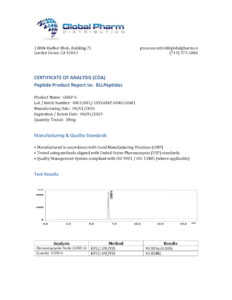
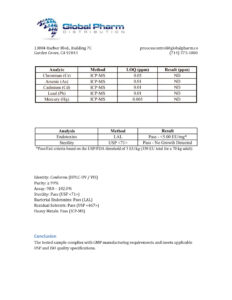
| Purity (Research Grade) | Greater than 98% by HPLC |
Structural Comparison with GHRP-2 and Ghrelin
GHRP-6 and GHRP-2 share four of six residues (Ala3, Trp4, D-Phe5, Lys6) with identical stereochemistry. The two N-terminal positions differ: GHRP-6 contains His1-D-Trp2, while GHRP-2 contains D-Ala1-D-2-Nal2. The replacement of histidine with D-alanine removed a protonatable aromatic group, while the substitution of D-tryptophan with D-2-naphthylalanine introduced a bicyclic naphthalene ring system that provides approximately 40% greater hydrophobic surface area compared to the monocyclic indole ring. These substitutions are responsible for GHRP-2's enhanced receptor binding affinity and greater GH-releasing potency [9].
GHRP-6 bears no primary sequence homology with ghrelin, the endogenous 28-amino acid acylated peptide ligand of GHS-R1a. Ghrelin requires an unusual octanoyl (eight-carbon fatty acid) modification on its Ser3 residue for receptor activation, whereas GHRP-6 achieves GHS-R1a engagement through its aromatic D-Trp2/Trp4 pharmacophore [4]. Despite these structural differences, both GHRP-6 and ghrelin bind to the same orthosteric site on GHS-R1a. Computational modeling studies suggest that the D-Trp2 indole ring of GHRP-6 occupies a spatial position analogous to the octanoyl chain of ghrelin, while the Trp4 indole maps to a position overlapping with ghrelin's Phe4 side chain [12]. This convergent pharmacophore occupancy explains why GHRP-6 and ghrelin compete for receptor binding in radioligand displacement assays.
Chemical Stability
GHRP-6 demonstrates good chemical stability in lyophilized form when stored at -20C, with shelf lives typically exceeding 24 months under appropriate conditions. In aqueous solution, GHRP-6 is susceptible to oxidation of the tryptophan indole rings (both D-Trp2 and Trp4), which can generate N-formylkynurenine and kynurenine derivatives. This oxidative degradation can be minimized by reconstitution in degassed, acidified water (pH 4-5), storage at 2-8C, and protection from light. The two D-amino acid residues confer significant resistance to enzymatic degradation by aminopeptidases and endopeptidases, but the L-amino acid residues (His1, Ala3, Trp4) remain susceptible to proteolysis in biological fluids, contributing to GHRP-6's relatively short plasma half-life of approximately 15-20 minutes following intravenous administration [8].
Detailed Mechanism of Action
GHS-R1a Receptor Activation
GHRP-6's primary mechanism of action involves agonism of the growth hormone secretagogue receptor type 1a (GHS-R1a), a seven-transmembrane G protein-coupled receptor identified by Howard and colleagues in 1996 [3]. GHS-R1a is expressed predominantly on somatotroph cells of the anterior pituitary gland, with additional expression on hypothalamic arcuate nucleus neurons, vagal afferent neurons, hippocampal neurons, and various peripheral tissues including the myocardium, gastrointestinal mucosa, pancreatic islets, and adrenal glands.
The signaling cascade initiated by GHRP-6 at pituitary somatotrophs proceeds through the following steps:
-
Receptor binding: GHRP-6 engages the orthosteric binding pocket of GHS-R1a within the transmembrane helical bundle. The D-Trp2 indole ring forms hydrophobic stacking interactions with aromatic residues in transmembrane helices 6 and 7, while the protonated Lys6 epsilon-amino group forms a salt bridge with Glu124 in transmembrane helix 3 [12]. The bis-indole framework of D-Trp2 and Trp4 provides the primary pharmacophoric contacts for receptor engagement.
-
Gq/11 coupling and PLC activation: Ligand binding induces a conformational change in GHS-R1a that promotes coupling to Gq/11 heterotrimeric G proteins. Activated Gq/11 stimulates phospholipase C-beta (PLC-beta), which hydrolyzes phosphatidylinositol 4,5-bisphosphate (PIP2) into inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG) [13].
-
Intracellular calcium mobilization: IP3 binds to IP3 receptors on the endoplasmic reticulum, triggering release of stored calcium into the cytoplasm. DAG concurrently activates protein kinase C (PKC), which phosphorylates voltage-gated L-type calcium channels at the plasma membrane, producing additional calcium influx from the extracellular space.
-
GH vesicle exocytosis: The combined calcium release from intracellular stores and extracellular influx produces a robust elevation in cytoplasmic calcium that triggers SNARE-mediated fusion of GH-containing secretory granules with the somatotroph plasma membrane, resulting in pulsatile GH release into the pituitary portal circulation [13].
Hypothalamic Actions
In addition to its direct pituitary effects, GHRP-6 exerts significant actions at the hypothalamic level. GHS-R1a is densely expressed on neuropeptide Y (NPY) and agouti-related peptide (AgRP) neurons in the arcuate nucleus, as well as on GHRH-producing neurons of the arcuate and ventromedial nuclei. GHRP-6 activates these hypothalamic populations to produce several complementary effects:
-
GHRH release stimulation: GHRP-6 activates GHS-R1a on GHRH neurons, stimulating the release of endogenous GHRH into the hypophyseal portal circulation. This GHRH then acts on pituitary GHRH receptors (a Gs/cAMP-coupled pathway), synergizing with GHRP-6's direct Gq/calcium signaling to produce amplified GH release. Dickson, Leng, and Robinson demonstrated that systemic administration of GHRP-6 activated c-fos expression in hypothalamic arcuate neurons, confirming central nervous system engagement [14].
-
Somatostatin modulation: Evidence from hypothalamic explant studies indicates that GHRP-6 can functionally antagonize somatostatin (SRIF) release from periventricular somatostatinergic neurons, effectively releasing the somatotroph from tonic inhibitory control. This disinhibition provides an additional amplification mechanism for GH release [15].
-
Potent appetite stimulation: Among the most prominent pharmacological effects of GHRP-6 is its ability to stimulate appetite and food intake. This effect is mediated through activation of GHS-R1a on arcuate NPY/AgRP orexigenic neurons, which increases the expression and release of these potent appetite-stimulating neuropeptides [16]. GHRP-6 produces notably stronger appetite stimulation than GHRP-2 and dramatically stronger effects than ipamorelin, which produces negligible appetite stimulation. The appetite-stimulating property of GHRP-6 directly mirrors the orexigenic action of endogenous ghrelin, consistent with their shared GHS-R1a agonism.
Synergy with GHRH Pathway
A critical pharmacological property of GHRP-6 is its profound synergistic interaction with GHRH signaling. When GHRP-6 and GHRH (or GHRH analogs) are co-administered, the resulting GH response is substantially greater than the sum of individual responses. Cheng and colleagues demonstrated this synergy in rat primary pituitary cell cultures, showing that GHRP-6 potentiated GHRH-stimulated GH release and intracellular cAMP accumulation in a manner consistent with convergent signaling through two distinct second messenger systems [17]:
-
GHRH pathway: GHRH-R activation leads to Gs-mediated stimulation of adenylyl cyclase, cAMP elevation, protein kinase A (PKA) activation, and PKA-dependent phosphorylation of L-type calcium channels and CREB transcription factor.
-
GHRP-6 pathway: GHS-R1a activation leads to Gq/11-mediated PLC activation, IP3-dependent calcium release, and PKC-dependent potentiation of calcium channels.
The simultaneous activation of cAMP/PKA and calcium/PKC signaling pathways produces a multiplicative effect on calcium-dependent exocytosis. Popovic and colleagues confirmed this synergy in human subjects, demonstrating that combined GHRP-6 plus GHRH produced GH peaks that were 2-3 fold greater than either agent alone [18]. Importantly, they also showed that this synergy was absent in patients with hypothalamic-pituitary disconnection, indicating that the hypothalamic component of GHRP-6's action is essential for full synergistic amplification [18].
Cortisol and Prolactin Co-Release
Unlike the highly selective ipamorelin, GHRP-6 produces substantial elevations in both cortisol and prolactin at GH-effective doses. The cortisol response is mediated through stimulation of adrenocorticotropic hormone (ACTH) release from anterior pituitary corticotrophs, likely through GHS-R1a expressed on these cells or through hypothalamic corticotropin-releasing hormone (CRH) neuron activation [2]. The prolactin response is thought to involve activation of lactotroph GHS-R1a or indirect modulation through hypothalamic dopaminergic pathways.
Bowers characterized the endocrine profile of GHRP-6 in comparative studies, demonstrating that at equi-effective GH-releasing doses, GHRP-6 produced relatively larger cortisol and prolactin elevations compared to GHRP-2, and dramatically larger elevations compared to ipamorelin [2]. This broader endocrine activation profile reflects the first-generation, less optimized nature of the GHRP-6 scaffold. Cordido and colleagues reported that in obese subjects, the combined administration of GHRP-6 and GHRH produced massive GH discharges, indicating that the somatotroph secretory reserve remains intact in obesity even though spontaneous GH secretion is reduced [19].
Somatostatin Sensitivity
Like other peptidyl GH secretagogues, GHRP-6-stimulated GH release remains sensitive to somatostatin (SRIF) inhibition. When somatostatin tone is elevated — following a GH pulse or during exogenous somatostatin infusion — GHRP-6's efficacy is attenuated. This somatostatin sensitivity preserves the pulsatile pattern of GH secretion that is essential for normal physiological responses, including hepatic IGF-1 production, linear bone growth, and metabolic regulation [15]. This property is shared with GHRP-2 and ipamorelin, and contrasts with the non-peptidyl GHS MK-677, which partially overrides somatostatin inhibition, potentially leading to more continuous (rather than pulsatile) GH elevation.
Constitutive GHS-R1a Activity and Inverse Agonism
An important aspect of GHS-R1a pharmacology, elucidated in part through studies with GHRP-6 and related compounds, is the receptor's high constitutive (ligand-independent) signaling activity. Holst and colleagues demonstrated that GHS-R1a exhibits approximately 50% of its maximal signaling activity in the absence of any agonist, making it one of the most constitutively active GPCRs known [12]. This basal activity contributes to tonic regulation of appetite and energy homeostasis. GHRP-6, as a full agonist, enhances signaling above this constitutive baseline, whereas certain inverse agonists can suppress it below baseline. Understanding this constitutive activity has been critical for interpreting the pharmacology of all GHS-R1a ligands, including GHRP-6.
Scientific Research Review
1. Growth Hormone Secretion and Neuroendocrine Pharmacology
GHRP-6 was the primary pharmacological tool used to characterize the GH secretagogue signaling pathway in the decades before ghrelin's discovery. Bowers' original studies in 1984 demonstrated that GHRP-6 produced dose-dependent GH release from rat anterior pituitary cells in vitro, with a maximal efficacy comparable to GHRH but operating through a mechanistically distinct, cAMP-independent pathway [1]. In vivo studies in humans confirmed potent GH-releasing activity following both intravenous and subcutaneous administration. Penalva and colleagues demonstrated that GHRP-6 at 1 microgram/kg IV produced GH peaks of 25-60 micrograms/L in healthy adults, with the response being highly reproducible and dose-dependent [20].
A particularly important finding was the demonstration by Popovic and colleagues that GHRP-6's GH-releasing effect in humans requires intact hypothalamic-pituitary connections. In patients with hypothalamic-pituitary disconnection (caused by pituitary stalk transection or large sellar/suprasellar tumors), GHRP-6-stimulated GH release was dramatically blunted or abolished, and the synergy between GHRP-6 and GHRH was completely lost [18]. This finding established that GHRP-6 acts primarily at the hypothalamic level in vivo, stimulating endogenous GHRH release and suppressing somatostatin, rather than acting exclusively through direct pituitary stimulation.
2. Appetite and Feeding Behavior Research
GHRP-6 is the most potently orexigenic compound among the classical GHRPs, making it a valuable research tool for studying appetite regulation and feeding behavior. The appetite-stimulating effect of GHRP-6 is mediated through GHS-R1a activation on hypothalamic NPY/AgRP neurons and is mechanistically identical to the orexigenic action of endogenous ghrelin [16]. Tschop, Smiley, and Heiman demonstrated in landmark studies that ghrelin and ghrelin-mimetic compounds including GHRP-6 induce adiposity in rodents, promoting food intake and fat deposition through a centrally mediated mechanism that is independent of GH release [16].
Intracerebroventricular (ICV) administration of GHRP-6 in rats produces robust, rapid increases in food intake that can be blocked by GHS-R1a antagonists and by NPY receptor antagonists, confirming the orexigenic pathway involves NPY/AgRP signaling downstream of GHS-R1a [21]. This appetite-stimulating property, while considered a limitation in certain research contexts, makes GHRP-6 a uniquely valuable tool for investigating the central mechanisms of hunger, satiety, and energy homeostasis. It has been used extensively in studies of feeding circuit neuroanatomy, the gut-brain axis, and the pathophysiology of cachexia and anorexia.
3. Gastroprotective Properties
One of the most significant and unexpected research findings with GHRP-6 is its potent gastroprotective activity. Gonzalez-Zulueta and colleagues, working at the Center for Genetic Engineering and Biotechnology (CIGB) in Havana, Cuba, demonstrated that GHRP-6 administration protects the gastric mucosa against injury induced by a variety of noxious agents, including ethanol, indomethacin, and ischemia-reperfusion [5]. This gastroprotective effect was shown to be independent of GH release, as it persisted in hypophysectomized animals, and was instead mediated through direct cytoprotective actions on gastric mucosal cells.
Subsequent studies by the same group and others revealed that GHRP-6's gastroprotective mechanism involves multiple pathways: upregulation of heat shock proteins (particularly HSP70), stimulation of nitric oxide (NO) production through endothelial nitric oxide synthase (eNOS) activation, reduction of pro-inflammatory cytokine expression (TNF-alpha, IL-1beta), and enhancement of mucosal blood flow [5, 22]. Berlanga and colleagues further demonstrated that GHRP-6 accelerated the healing of established gastric ulcers in rat models, promoting angiogenesis and epithelial restitution at the ulcer margin [22]. These findings have positioned GHRP-6 as a leading research compound for investigating peptide-mediated cytoprotection in the gastrointestinal tract.
4. Cardioprotective Research
GHRP-6 has demonstrated significant cardioprotective properties in preclinical models of myocardial ischemia-reperfusion injury. Studies by the CIGB group showed that systemic administration of GHRP-6 prior to coronary artery ligation in rats reduced infarct size by approximately 40-60% compared to vehicle-treated controls [6]. This cardioprotection was associated with reduced oxidative stress, decreased cardiomyocyte apoptosis, and preservation of mitochondrial membrane potential.
The cardioprotective mechanism of GHRP-6 appears to involve activation of the reperfusion injury salvage kinase (RISK) pathway, including PI3K/Akt and ERK1/2 signaling cascades, as well as upregulation of anti-apoptotic Bcl-2 family members and heat shock proteins in cardiomyocytes [6]. Importantly, these cardioprotective effects appear to be mediated through GHS-R1a expressed on cardiomyocytes rather than through GH release, as the protective effects persisted in the presence of GH receptor blockade. These findings parallel observations with ghrelin itself, which has been shown by Nagaya and colleagues to improve cardiac function in patients with chronic heart failure [23], and suggest that the GHS-R1a/ghrelin system plays an important role in myocardial stress responses.
5. Anti-Fibrotic and Wound Healing Research
Building on the cytoprotective findings in the gastrointestinal tract and heart, researchers have investigated GHRP-6's effects on tissue fibrosis and wound healing. Studies by Berlanga-Acosta and colleagues demonstrated that GHRP-6 reduces liver fibrosis in carbon tetrachloride-treated rats, decreasing collagen deposition, hepatic stellate cell activation, and expression of the pro-fibrotic cytokine transforming growth factor-beta (TGF-beta) [22]. The anti-fibrotic mechanism was associated with downregulation of connective tissue growth factor (CTGF) and upregulation of matrix metalloproteinases (MMPs) that degrade excess extracellular matrix.
In wound healing models, GHRP-6 accelerated closure of full-thickness skin wounds in diabetic mice, a model characterized by impaired wound healing. The mechanism involved enhanced angiogenesis, increased expression of vascular endothelial growth factor (VEGF) and epidermal growth factor (EGF), and modulation of the inflammatory response to favor tissue repair over chronic inflammation [22]. These findings suggest that GHRP-6's cytoprotective properties extend beyond the acute phase of injury to encompass tissue remodeling and regeneration.
6. Neuroprotective Research
GHRP-6 has demonstrated neuroprotective properties in preclinical models of cerebral ischemia and neurodegeneration. Frago, Paneda, and colleagues showed that GHRP-6 increases brain insulin-like growth factor-I (IGF-I) expression and activates intracellular signaling pathways involved in neuronal survival, including the PI3K/Akt pathway, in rat brain tissue [24]. Systemic administration of GHRP-6 produced significant increases in brain IGF-I mRNA and protein levels, suggesting that the peptide can influence central nervous system trophic factor expression through GHS-R1a-mediated signaling.
In models of cerebral ischemia, GHRP-6 pretreatment reduced infarct volume, decreased neuronal apoptosis, and attenuated microglial activation and neuroinflammation [24]. These neuroprotective effects were associated with activation of anti-apoptotic signaling (Bcl-2, Bcl-XL) and suppression of pro-apoptotic mediators (Bax, caspase-3). The presence of GHS-R1a on hippocampal neurons and cortical interneurons provides the anatomical substrate for these direct central neuroprotective actions.
7. GHS-R1a Receptor Pharmacology and Drug Discovery
As the founding GHS compound, GHRP-6 has served as a critical reference agonist for GHS-R1a receptor pharmacology. It is routinely used in radioligand binding assays, functional calcium mobilization assays, beta-arrestin recruitment assays, and structure-activity relationship campaigns aimed at developing novel GHS-R1a modulators [3, 12]. The well-characterized pharmacological profile of GHRP-6 makes it an indispensable positive control for receptor screening, and its distinct endocrine signature (strong GH release with appetite stimulation and cortisol/prolactin co-release) provides a benchmark for evaluating the selectivity improvements achieved by newer compounds.
GHRP-6 was also instrumental in elucidating the constitutive activity of GHS-R1a. Studies comparing the effects of GHRP-6 (a full agonist), partial agonists, and inverse agonists at GHS-R1a revealed the receptor's unusually high basal signaling activity, which has profound implications for understanding energy homeostasis and has guided the development of inverse agonist compounds as potential anti-obesity therapeutics [12].
Comparison with Other GH Secretagogues
GHRP Family Comparison
| Property | GHRP-6 | GHRP-2 | Ipamorelin | Hexarelin |
|---|---|---|---|---|
| Sequence | His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 | D-Ala-D-2Nal-Ala-Trp-D-Phe-Lys-NH2 | Aib-His-D-2Nal-D-Phe-Lys-NH2 | His-D-2MeTrp-Ala-Trp-D-Phe-Lys-NH2 |
| Residue Count | 6 (hexapeptide) | 6 (hexapeptide) | 5 (pentapeptide) | 6 (hexapeptide) |
| Molecular Weight | Approximately 873.01 Da | Approximately 817.97 Da | Approximately 711.85 Da | Approximately 887.04 Da |
| GH Release Potency | Moderate-High | High (reference standard) | Moderate | High |
| GH Selectivity | Low | Moderate | Very High | Low-Moderate |
| Appetite Stimulation | Strong | Moderate | Minimal | Moderate |
| Cortisol Elevation | Moderate-High | Moderate | Negligible | Moderate |
| Prolactin Elevation | Moderate | Mild-Moderate | Negligible | Moderate |
| Primary Research Use | Ghrelin mimetic research, gastroprotection | Diagnostic (Japan), GH axis research | Selective GH studies, bone research | Cardiac research, GH studies |
| Generation | First | Second | Third | Second |
Mechanistic Comparison: Peptide vs. Non-Peptide GHS
| Feature | GHRP-6 (Peptide GHS) | MK-677 / Ibutamoren (Non-Peptide GHS) |
|---|---|---|
| Receptor | GHS-R1a | GHS-R1a |
| Administration | Injectable (IV, SC) | Oral |
| Plasma Half-Life | Approximately 15-20 minutes | Approximately 4-6 hours |
| GH Release Pattern | Pulsatile | More sustained/tonic |
| Somatostatin Sensitivity | Sensitive (preserves pulsatility) | Partially resistant |
| Appetite Stimulation | Strong, acute | Moderate, sustained |
| Cortisol/Prolactin Effects | Present at GH-effective doses | Present at GH-effective doses |
| Duration of GH Elevation | Approximately 1-2 hours | Approximately 8-12 hours |
| Oral Bioavailability | Negligible | Approximately 60% |
GHRP-6 vs. GHRH Analogs
| Feature | GHRP-6 | GHRH Analogs (Sermorelin, CJC-1295) |
|---|---|---|
| Receptor Target | GHS-R1a (Gq/11 coupled) | GHRH-R (Gs coupled) |
| Second Messenger | IP3/DAG/Calcium | cAMP/PKA |
| Hypothalamic Action | Stimulates GHRH release, suppresses somatostatin | Direct pituitary action (primarily) |
| Appetite Effects | Strong stimulation | None |
| Cortisol Effects | Elevates cortisol | No effect on cortisol |
| Synergy | Synergizes with GHRH | Synergizes with GHRPs |
| GH Deficiency Diagnosis | Blunted in hypothalamic disease | Blunted in pituitary disease |
Pharmacokinetics and Safety Profile
Pharmacokinetics
GHRP-6 exhibits pharmacokinetic properties typical of small peptides in systemic circulation. Following intravenous administration, GHRP-6 produces rapid peak plasma concentrations with a distribution half-life of approximately 2-5 minutes and an elimination half-life of approximately 15-20 minutes [8]. The short half-life reflects rapid enzymatic degradation by circulating peptidases and renal clearance.
Subcutaneous administration produces somewhat delayed peak plasma concentrations (Tmax approximately 15-30 minutes) with a lower Cmax but extended duration of exposure compared to IV administration. The subcutaneous bioavailability of GHRP-6 has not been precisely determined in published literature but is estimated at approximately 60-80% based on GH response comparisons between IV and SC routes [8]. Oral bioavailability is negligible due to extensive first-pass metabolism and degradation by gastrointestinal proteases, although intranasal administration has been explored as an alternative non-injectable route with limited success.
The peak GH response following GHRP-6 administration typically occurs 15-30 minutes after IV injection and 30-60 minutes after SC injection, with GH levels returning to baseline within approximately 2-3 hours. The rapid onset and short duration of action support a pulsatile administration pattern that aligns with the physiological pattern of GH secretion.
Factors Affecting GH Response
The magnitude of the GH response to GHRP-6 is influenced by several physiological and pharmacological variables:
-
Age: GH responses to GHRP-6 are generally larger in younger subjects and decline with advancing age, reflecting age-related decreases in somatotroph mass and GHS-R1a expression. However, GHRP-6 retains significant GH-releasing efficacy in elderly subjects, particularly when combined with GHRH [20].
-
Body composition: Obesity is associated with reduced GH responses to GHRP-6, consistent with the well-documented somatotroph suppression in obesity. However, Cordido and colleagues demonstrated that the combination of GHRP-6 plus GHRH can produce massive GH responses even in obese subjects, indicating that somatotroph secretory capacity is preserved but tonically inhibited [19].
-
Sex steroids: Estrogen enhances GH responses to GHRP-6, which may contribute to the generally larger GH pulses observed in premenopausal women compared to age-matched men. This effect is thought to involve estrogen-mediated upregulation of GHS-R1a expression on somatotrophs [13].
-
Somatostatin tone: Elevated somatostatin signaling (as occurs in the trough between GH pulses) attenuates GHRP-6-stimulated GH release. Conversely, withdrawal of somatostatin tone (as occurs at the onset of a GH pulse) amplifies the response [15].
-
Prior GHRP-6 exposure: Repeated administration of GHRP-6 may produce partial tachyphylaxis (attenuation of response), particularly with frequent dosing. This desensitization is thought to involve GHS-R1a internalization and downregulation following sustained agonist exposure [13].
Safety Profile
The safety profile of GHRP-6 has been characterized through decades of preclinical research and limited human studies. The following effects have been consistently observed:
Common effects (expected pharmacological actions):
- Increased appetite and food intake (ghrelin-mimetic effect)
- Transient increases in serum cortisol (typically within physiological range)
- Transient increases in serum prolactin (typically within physiological range)
- Transient facial flushing following injection (particularly IV)
- Mild, transient water retention
Less common effects:
- Mild gastrointestinal disturbance (nausea at high doses)
- Transient tingling or numbness at injection site
- Dizziness or lightheadedness (associated with GH-mediated vasodilation)
Endocrine considerations:
- The cortisol elevation produced by GHRP-6 is consistently greater than that produced by GHRP-2 and substantially greater than that produced by ipamorelin at equi-effective GH-releasing doses [2]. While these elevations remain within physiological ranges in most studies, the potential for HPA axis stimulation should be considered in research protocols, particularly those involving repeated administration.
- The prolactin elevation, while typically modest and transient, distinguishes GHRP-6 from the prolactin-neutral profile of ipamorelin.
- The strong appetite-stimulating effect of GHRP-6 is a primary pharmacological action rather than a side effect, directly reflecting ghrelin-mimetic activity at hypothalamic GHS-R1a.
Long-term safety data:
Published long-term safety data for GHRP-6 are limited compared to GHRP-2 (which has regulatory approval in Japan). Bowers and colleagues conducted a 30-day continuous subcutaneous infusion study of GHRP-2 (not GHRP-6) in elderly subjects that provided sustained GH elevation without significant adverse events [25], and the closely related pharmacology of GHRP-6 suggests a comparable safety margin. However, dedicated long-term safety studies of GHRP-6 in humans remain an area requiring further investigation.
Research Applications
1. GHS-R1a/Ghrelin System Pharmacology
GHRP-6 remains an essential reference compound for studying the GHS-R1a receptor and the broader ghrelin signaling system. As the original synthetic GHS-R1a agonist, GHRP-6 is used as a positive control in receptor binding assays, calcium mobilization assays, and beta-arrestin recruitment screens. Its well-characterized pharmacological profile makes it indispensable for validating new assay systems, evaluating novel GHS-R1a ligands, and conducting comparative pharmacology studies across the GHS compound family [3, 12].
2. Appetite and Energy Homeostasis Research
GHRP-6's potent orexigenic activity makes it a leading tool for investigating the central mechanisms of appetite regulation, feeding behavior, and energy balance. Researchers use GHRP-6 to activate orexigenic circuits in animal models, study the neuroanatomy and neurochemistry of hunger signaling, and investigate the pathophysiology of conditions characterized by altered appetite, including cachexia, anorexia nervosa, and obesity [16, 21].
3. Gastroprotection and Gastrointestinal Research
The discovery of GHRP-6's potent gastroprotective properties has established it as a leading research compound for investigating peptide-mediated mucosal cytoprotection. GHRP-6 is used in models of gastric ulceration, NSAID-induced gastropathy, and ischemia-reperfusion injury of the gastrointestinal tract, with potential implications for understanding how the ghrelin system protects the gut mucosa under stress conditions [5, 22].
4. Cardioprotection and Cardiovascular Research
GHRP-6's cardioprotective effects in ischemia-reperfusion models have established its utility for investigating myocardial stress responses and the role of GHS-R1a signaling in cardiac protection. Research applications include studying the RISK pathway in cardiomyocytes, investigating the relationship between ghrelin signaling and cardiac remodeling, and evaluating the therapeutic potential of GHS-R1a agonism in heart failure and ischemic heart disease [6, 23].
5. Anti-Fibrotic and Tissue Repair Research
GHRP-6's anti-fibrotic effects in liver and skin models position it as a research tool for investigating peptide-mediated anti-fibrotic mechanisms. Applications include studying hepatic stellate cell biology, the role of TGF-beta/CTGF signaling in fibrogenesis, and the potential for GHS-R1a agonism to promote tissue repair in chronic injury states [22].
6. Neuroendocrine Diagnostics Research
Although GHRP-6 has not achieved the regulatory approval obtained by GHRP-2 (pralmorelin) in Japan, it has been extensively studied as a diagnostic tool for GH deficiency. The GHRP-6 stimulation test, often combined with GHRH, has been investigated as an alternative to the insulin tolerance test (ITT) for assessing pituitary GH reserve. Popovic and colleagues demonstrated that the combined GHRP-6 plus GHRH test can distinguish between hypothalamic and pituitary causes of GH deficiency based on the presence or absence of synergistic GH release [18].
7. Neuroprotection Research
GHRP-6's ability to increase brain IGF-I expression and activate neuroprotective signaling pathways has positioned it as a tool for studying the role of GHS-R1a in neuronal survival and brain injury recovery. Research applications include models of cerebral ischemia, traumatic brain injury, and age-related neurodegeneration [24].
8. Combination Peptide Research
GHRP-6 is frequently used in combination with GHRH analogs to study the synergistic amplification of GH release. These combination protocols exploit the convergence of Gq/calcium and Gs/cAMP signaling pathways on the somatotroph to achieve GH responses that exceed those attainable with any single agent. Such protocols are used to study maximal pituitary capacity, investigate the physiological ceiling of GH secretion, and develop optimized research paradigms [17, 18, 19].
9. Anti-Doping and Analytical Chemistry
Due to its potent GH-releasing properties, GHRP-6 has been included on the World Anti-Doping Agency (WADA) prohibited list as a growth hormone releasing factor. This has stimulated significant research into analytical detection methods for GHRP-6 and its metabolites in biological matrices, advancing mass spectrometry-based techniques for peptide detection in urine and blood samples [13].
References
[1] Bowers, C.Y., Momany, F.A., Reynolds, G.A., & Hong, A. (1984). "On the in vitro and in vivo activity of a new synthetic hexapeptide that acts on the pituitary to specifically release growth hormone." Endocrinology, 114(5), 1537-1545. DOI: 10.1210/endo-114-5-1537
[2] Bowers, C.Y. (1998). "Growth hormone-releasing peptide (GHRP)." Cellular and Molecular Life Sciences, 54(12), 1316-1329. DOI: 10.1007/s000180050257
[3] Howard, A.D., Feighner, S.D., Cully, D.F., et al. (1996). "A receptor in pituitary and hypothalamus that functions in growth hormone release." Science, 273(5277), 974-977. DOI: 10.1126/science.273.5277.974
[4] Kojima, M., Hosoda, H., Date, Y., Nakazato, M., Matsuo, H., & Kangawa, K. (1999). "Ghrelin is a growth-hormone-releasing acylated peptide from stomach." Nature, 402(6762), 656-660. DOI: 10.1038/45230
[5] Gonzalez-Zulueta, M., Berlanga, J., Leyva, A., et al. (2007). "Growth hormone-releasing peptide 6 (GHRP-6) prevents ethanol-induced gastric mucosal damage in rats." Growth Hormone & IGF Research, 17(3), 236-242. DOI: 10.1016/j.ghir.2007.01.010
[6] Berlanga, J., Cibrian, D., Guevara, L., et al. (2007). "Growth-hormone-releasing peptide 6 (GHRP-6) prevents oxidant cytotoxicity and reduces myocardial necrosis in a model of acute myocardial infarction." Clinical Science, 112(4), 241-250. DOI: 10.1042/CS20060185
[7] Momany, F.A., Bowers, C.Y., Reynolds, G.A., et al. (1981). "Design, synthesis, and biological activity of peptides which release growth hormone in vitro." Endocrinology, 108(1), 31-39. DOI: 10.1210/endo-108-1-31
[8] Bowers, C.Y., Momany, F., Reynolds, G.A., et al. (1980). "Structure-activity relationships of a synthetic pentapeptide that specifically releases growth hormone in vitro." Endocrinology, 106(3), 663-667. DOI: 10.1210/endo-106-3-663
[9] Arvat, E., Maccario, M., Di Vito, L., et al. (2001). "Endocrine activities of ghrelin, a natural growth hormone secretagogue (GHS), in humans: Comparison and interactions with hexarelin, a nonnatural peptidyl GHS, and GH-releasing hormone." Journal of Clinical Endocrinology and Metabolism, 86(3), 1169-1174. DOI: 10.1210/jcem.86.3.7314
[10] Raun, K., Hansen, B.S., Johansen, N.L., et al. (1998). "Ipamorelin, the first selective growth hormone secretagogue." European Journal of Endocrinology, 139(5), 552-561. DOI: 10.1530/eje.0.1390552
[11] Bowers, C.Y. (2001). "Unnatural growth hormone-releasing peptide begets natural ghrelin." Journal of Clinical Endocrinology and Metabolism, 86(4), 1464-1469. DOI: 10.1210/jcem.86.4.7399
[12] Holst, B., Cygankiewicz, A., Jensen, T.H., Ankersen, M., & Schwartz, T.W. (2003). "High constitutive signaling of the ghrelin receptor — identification of a potent inverse agonist." Molecular Endocrinology, 17(11), 2201-2210. DOI: 10.1210/me.2003-0069
[13] Ghigo, E., Arvat, E., Muccioli, G., & Camanni, F. (1997). "Growth hormone-releasing peptides." European Journal of Endocrinology, 136(5), 445-460. DOI: 10.1530/eje.0.1360445
[14] Dickson, S.L., Leng, G., & Robinson, I.C.A.F. (1993). "Systemic administration of growth hormone-releasing peptide activates hypothalamic arcuate neurons." Neuroscience, 53(2), 303-306. DOI: 10.1016/0306-4522(93)90197-N
[15] Tannenbaum, G.S. & Bowers, C.Y. (2001). "Interactions of growth hormone secretagogues and growth hormone-releasing hormone/somatostatin." Endocrine, 14(1), 21-27. DOI: 10.1385/ENDO:14:1:021
[16] Tschop, M., Smiley, D.L., & Heiman, M.L. (2000). "Ghrelin induces adiposity in rodents." Nature, 407(6806), 908-913. DOI: 10.1038/35038090
[17] Cheng, K., Chan, W.W., Barreto, A., Convey, E.M., & Smith, R.G. (1989). "The synergistic effects of His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 on growth hormone (GH)-releasing factor-stimulated GH release and intracellular adenosine 3',5'-monophosphate accumulation in rat primary pituitary cell culture." Endocrinology, 124(6), 2791-2798. DOI: 10.1210/endo-124-6-2791
[18] Popovic, V., Damjanovic, S., Micic, D., et al. (1995). "Blocked growth hormone-releasing peptide (GHRP-6)-induced GH secretion and absence of the synergic action of GHRP-6 plus GH-releasing hormone in patients with hypothalamopituitary disconnection: Evidence that GHRP-6 main action is exerted at the hypothalamic level." Journal of Clinical Endocrinology and Metabolism, 80(3), 942-947. DOI: 10.1210/jcem.80.3.7883853
[19] Cordido, F., Penalva, A., Dieguez, C., & Casanueva, F.F. (1993). "Massive growth hormone (GH) discharge in obese subjects after the combined administration of GH-releasing hormone and GHRP-6: Evidence for a marked somatotroph secretory capability in obesity." Journal of Clinical Endocrinology and Metabolism, 76(4), 819-823. DOI: 10.1210/jcem.76.4.8473388
[20] Penalva, A., Carballo, A., Pombo, M., Casanueva, F.F., & Dieguez, C. (1993). "Effect of growth hormone (GH)-releasing hormone (GHRH), atropine, pyridostigmine, or hypoglycemia on GHRP-6-induced GH secretion in man." Journal of Clinical Endocrinology and Metabolism, 76(1), 168-171. DOI: 10.1210/jcem.76.1.8421085
[21] Wren, A.M., Small, C.J., Abbott, C.R., et al. (2001). "Ghrelin causes hyperphagia and obesity in rats." Diabetes, 50(11), 2540-2547. DOI: 10.2337/diabetes.50.11.2540
[22] Berlanga-Acosta, J., Abreu-Cruz, A., Herrera, D.G.B., et al. (2009). "Synthetic growth hormone-releasing peptides (GHRPs): A historical appraisal of the evidences supporting their cytoprotective effects." Clinical Medicine Insights: Cardiology, 3, 5-15. DOI: 10.4137/CMC.S2067
[23] Nagaya, N., Moriya, J., Yasumura, Y., et al. (2004). "Effects of ghrelin administration on left ventricular function, exercise capacity, and muscle wasting in patients with chronic heart failure." Circulation, 110(24), 3674-3679. DOI: 10.1161/01.CIR.0000149746.62908.BB
[24] Frago, L.M., Paneda, C., Dickson, S.L., Hewson, A.K., Argente, J., & Chowen, J.A. (2002). "Growth hormone (GH) and GH-releasing peptide-6 increase brain insulin-like growth factor-I expression and activate intracellular signaling pathways involved in neuroprotection." Endocrinology, 143(10), 4113-4122. DOI: 10.1210/en.2002-220167
[25] Bowers, C.Y., Granda, R., Mohan, S., Kuipers, J., Baylink, D., & Veldhuis, J.D. (2004). "Sustained elevation of pulsatile growth hormone (GH) secretion and insulin-like growth factor I (IGF-I), IGF-binding protein 3 (IGFBP-3), and IGFBP-5 concentrations during 30-day continuous subcutaneous infusion of GH-releasing peptide-2 in older men and women." Journal of Clinical Endocrinology and Metabolism, 89(5), 2290-2300. DOI: 10.1210/jc.2003-031799
Disclaimer
This article is for educational and informational purposes only. It is not intended as medical advice, diagnosis, or treatment recommendation. GHRP-6 is sold as a research peptide and is not approved for human therapeutic use by the FDA or any other major regulatory agency. The information presented herein is derived from published peer-reviewed scientific literature and does not constitute a recommendation for any specific research protocol or application. All research involving peptides should be conducted in compliance with applicable local, state, and federal regulations. Researchers should consult relevant institutional review boards and regulatory bodies before initiating any research protocols.
Published by BLL Peptides — Premium Research Peptides
GHRP-6 is a research-grade growth hormone releasing hexapeptide studied for GH secretion stimulation, appetite promotion, and tissue repair support through GH-dependent metabolic pathways. Researchers investigating the growth hormone axis and body composition rely on pharmaceutical-grade purity for valid experimental outcomes. Available at BLL Peptides — USA-made, rigorously tested.
| ✅ COA tested every batch | ✅ 98%+ purity guaranteed |
| ✅ USA manufactured, GMP-certified | ✅ Glass vials — not plastic |
| ✅ Veteran-owned company | ✅ Free shipping over $150 |