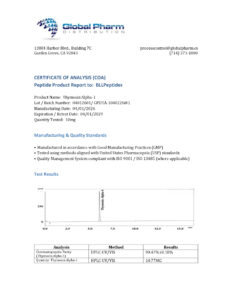
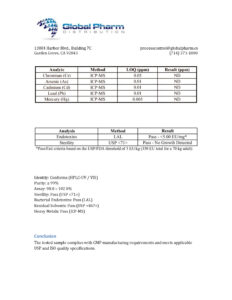
Thymosin Alpha-1 10mg (3ml)
Thymosin Alpha-1 is a research-grade immunomodulatory thymic peptide studied for T-cell activation, immune regulation, and antiviral response mechanisms. Researchers investigating immune system optimization and autoimmune pathways rely on pharmaceutical-grade purity for reliable, reproducible outcomes. Available at BLL Peptides — USA-made, rigorously tested. ✅ COA tested every batch✅ 98%+ purity guaranteed ✅ USA manufactured, GMP-certified✅ Glass…
Description
Thymosin Alpha-1: Complete Research Guide – Thymic Peptide Mechanisms, Immune Modulation Research, and Clinical Applications
Last updated: March 2026
Executive Summary
Thymosin Alpha-1 (Ta1) is a naturally occurring 28-amino acid peptide first isolated from thymosin fraction 5, a partially purified extract of the bovine thymus gland, by Allan Goldstein and colleagues at George Washington University in 1977. The peptide has since been recognized as one of the most clinically advanced immunomodulatory peptides in the world, with its synthetic form thymalfasin (marketed as Zadaxin) approved in over 35 countries for the treatment of chronic hepatitis B and C, and investigated extensively in cancer immunotherapy, immunodeficiency states, and vaccine adjuvant applications [1].
The primary sequence of Thymosin Alpha-1 is Ac-SDAAVDTSSEITTKDLKEKKEVVEEAEN, with a distinctive N-terminal acetylation on the serine residue that is critical for biological activity and metabolic stability. Its molecular formula is C129H215N33O55, with a molecular weight of approximately 3,108 Da (CAS: 62304-98-7). Despite its relatively small size, Ta1 exerts profound effects on both innate and adaptive immune function through activation of Toll-like receptors (TLR2 and TLR9), promotion of dendritic cell maturation, enhancement of T-cell differentiation and function, and stimulation of natural killer (NK) cell cytotoxicity [2].
Unlike many immunostimulatory agents that risk overactivation and cytokine storm, Thymosin Alpha-1 acts as a true immune modulator, restoring immune homeostasis by upregulating suppressed immune responses while simultaneously tempering excessive inflammatory signaling. This bidirectional regulatory capacity distinguishes Ta1 from conventional immunostimulants and underlies its favorable safety profile observed across decades of clinical use [3]. The peptide has demonstrated particular value in immunocompromised populations, including elderly patients with age-related immune decline (immunosenescence), individuals with chronic viral hepatitis, cancer patients undergoing chemotherapy, and subjects with primary or secondary immunodeficiencies.
This comprehensive guide examines the molecular structure, mechanisms of action, clinical and preclinical evidence base, safety profile, and research applications of Thymosin Alpha-1, providing researchers with a rigorous, evidence-based resource grounded in peer-reviewed literature. Connections to related thymic peptides, including Thymalin and the antimicrobial peptide LL-37, are discussed where mechanistically relevant.
Interactive Molecular Structure
The following interactive 3D visualization renders the Thymosin Alpha-1 peptide backbone in its alpha-helical conformation. The N-terminally acetylated Ser1 residue (orange) represents a hallmark structural feature essential for biological activity and resistance to aminopeptidase degradation. The peptide displays a characteristic abundance of negatively charged residues (Asp, Glu in orange-red) and positively charged lysine residues (red), reflecting its highly charged nature at physiological pH.
Legend: The interactive visualization above depicts the 28-residue alpha-helical backbone of Thymosin Alpha-1. The acetylated Ser1 (orange glow) at the N-terminus is a defining structural feature that protects the peptide from aminopeptidase degradation and is required for full biological activity. Negatively charged residues (Asp, Glu in orange-red) are abundant throughout the C-terminal half, contributing to a net negative charge at physiological pH. Positively charged lysine residues (red) cluster in the mid-region (positions 14, 17, 19, 20), creating a cationic patch implicated in receptor interactions. Dashed lines represent i-to-i+4 hydrogen bonds stabilizing the alpha-helix. Drag to rotate; scroll to zoom.
Table of Contents
- Introduction and Discovery History
- Molecular Structure and Chemistry
- Mechanism of Action
- Scientific Research Review
- Comparison with Related Immunomodulatory Peptides
- Safety Profile and Pharmacology
- Research Applications
- References
- Disclaimer
Introduction and Discovery History
The Thymus Gland and Thymic Hormone Research
The thymus gland has fascinated immunologists since its role in adaptive immunity was first established by Jacques Miller in 1961, who demonstrated that neonatal thymectomy in mice abolished the capacity for cell-mediated immune responses. This landmark finding established the thymus as the primary organ for T-lymphocyte maturation and ignited a decades-long search for the soluble factors responsible for thymic function [4].
By the late 1960s and early 1970s, multiple research groups were racing to isolate and characterize thymic hormones. Abraham White and Allan Goldstein at the Albert Einstein College of Medicine developed a methodology to extract a crude mixture of thymic polypeptides from calf thymus tissue, which they designated "thymosin fraction 5." This heterogeneous preparation contained approximately 40 distinct peptides ranging from 1,000 to 15,000 Da and demonstrated potent immunorestortive activity in animal models of immunodeficiency [5].
Thymosin fraction 5 was among the first thymic preparations to enter clinical trials, showing promise in restoring immune function in patients with primary immunodeficiencies and as an adjunct to cancer chemotherapy. However, the heterogeneous nature of the preparation made standardization difficult and prompted efforts to isolate and characterize the individual bioactive components.
Isolation of Thymosin Alpha-1
The isolation of Thymosin Alpha-1 from thymosin fraction 5 was achieved in 1977 by Allan Goldstein and colleagues, who had by then relocated to George Washington University. Using a combination of isoelectric focusing, gel filtration, and reverse-phase high-performance liquid chromatography, they purified a single 28-amino acid peptide that accounted for a substantial portion of the immunomodulatory activity of the parent fraction [1].
The complete amino acid sequence was determined as Ac-Ser-Asp-Ala-Ala-Val-Asp-Thr-Ser-Ser-Glu-Ile-Thr-Thr-Lys-Asp-Leu-Lys-Glu-Lys-Lys-Glu-Val-Val-Glu-Glu-Ala-Glu-Asn. Several features of this sequence were immediately notable. The N-terminal acetylation was unusual for a naturally occurring peptide of this size and suggested specific post-translational processing. The abundance of acidic residues (seven glutamate and three aspartate residues, totaling ten of 28 positions) gave the peptide a calculated isoelectric point of approximately 4.2, making it strongly anionic at physiological pH [6].
Goldstein's group demonstrated that synthetic Thymosin Alpha-1 reproduced the immunomodulatory activities of the natural peptide, confirming the structural assignment and opening the door to large-scale synthesis for research and clinical applications. The synthetic peptide, later designated thymalfasin, became the basis for the commercial product Zadaxin developed by SciClone Pharmaceuticals.
From Laboratory to Clinic: The Development of Zadaxin
The clinical development of Thymosin Alpha-1 proceeded through a path unusual in pharmaceutical history. Rather than following the traditional Western regulatory trajectory through the FDA, thymalfasin (Zadaxin) was developed primarily in Asian and European markets where the burden of chronic hepatitis B was highest. The first regulatory approvals came in the mid-1990s, with Zadaxin approved for the treatment of chronic hepatitis B in countries including China, the Philippines, and several nations in Latin America and Eastern Europe [7].
By 2010, Zadaxin had received regulatory approval in more than 35 countries for indications including chronic hepatitis B, chronic hepatitis C (as adjunctive therapy with interferon), and as an immune modulator in cancer patients. Notably, it remained unapproved by the U.S. FDA and the European Medicines Agency, not due to safety concerns but largely reflecting the complex regulatory landscape and the focus of clinical trials in markets with higher hepatitis prevalence.
The clinical journey of Thymosin Alpha-1 also intersected with the broader history of thymic peptide research. Thymalin, a related thymic extract developed in Russia by Vladimir Khavinson, represents a complementary approach to thymic immune restoration. While Thymalin contains a mixture of thymic peptides rather than a single defined molecule, both agents share the conceptual foundation of restoring age-related and disease-related immune decline through thymic peptide supplementation.
The COVID-19 Era and Renewed Interest
The SARS-CoV-2 pandemic beginning in 2020 brought renewed attention to Thymosin Alpha-1 as clinicians in China and other countries with existing Zadaxin availability began investigating its potential as an adjunctive therapy for COVID-19. Several observational studies and small clinical trials reported that Thymosin Alpha-1 administration was associated with improved lymphocyte counts and reduced mortality in critically ill COVID-19 patients, particularly those with lymphopenia [8]. While these findings were preliminary and subject to significant methodological limitations, they reinvigorated global interest in Thymosin Alpha-1 as an immune restorative agent and prompted calls for larger, randomized controlled trials.
Molecular Structure and Chemistry
Primary Structure and Sequence Analysis
Thymosin Alpha-1 consists of 28 amino acid residues with the following complete sequence:
Ac-Ser-Asp-Ala-Ala-Val-Asp-Thr-Ser-Ser-Glu-Ile-Thr-Thr-Lys-Asp-Leu-Lys-Glu-Lys-Lys-Glu-Val-Val-Glu-Glu-Ala-Glu-Asn
Position-by-position breakdown:
| Position | Residue | Character | Functional Significance |
|---|---|---|---|
| 1 | Ac-Ser | Acetylated/Polar | N-terminal acetylation; protease protection |
| 2 | Asp | Negative | Contributes to net negative charge |
| 3-4 | Ala-Ala | Hydrophobic | Helix-stabilizing residues |
| 5 | Val | Hydrophobic | Core hydrophobic contribution |
| 6 | Asp | Negative | Electrostatic interactions |
| 7 | Thr | Polar | Hydrogen bonding; helix N-cap |
| 8-9 | Ser-Ser | Polar | Flexible polar region |
| 10 | Glu | Negative | Charged surface residue |
| 11 | Ile | Hydrophobic | Hydrophobic core stabilization |
| 12-13 | Thr-Thr | Polar | Hydrogen bonding network |
| 14 | Lys | Positive | Cationic patch; receptor interaction |
| 15 | Asp | Negative | Potential salt bridge with Lys14 |
| 16 | Leu | Hydrophobic | Core hydrophobic contribution |
| 17 | Lys | Positive | Cationic patch; TLR interaction region |
| 18 | Glu | Negative | Salt bridge potential with Lys17 |
| 19-20 | Lys-Lys | Positive | Cationic cluster; critical for activity |
| 21 | Glu | Negative | Charged surface; solubility |
| 22-23 | Val-Val | Hydrophobic | Hydrophobic core |
| 24-25 | Glu-Glu | Negative | C-terminal acidic cluster |
| 26 | Ala | Hydrophobic | Helix stabilization |
| 27 | Glu | Negative | C-terminal charge |
| 28 | Asn | Polar | C-terminal amide hydrogen bonding |
Molecular properties:
| Property | Value |
|---|---|
| Molecular formula | C129H215N33O55 |
| Molecular weight | Approximately 3,108 Da |
| CAS number | 62304-98-7 |
| Isoelectric point (pI) | Approximately 4.2 |
| Net charge (pH 7.4) | Approximately -6 |
| N-terminal modification | Acetylation (Ac-Ser) |
N-Terminal Acetylation: A Critical Modification
The N-terminal acetylation of Thymosin Alpha-1 is not merely a biochemical curiosity but a functionally essential modification. Studies by Spangelo and colleagues demonstrated that removal of the acetyl group significantly reduced the immunomodulatory activity of the peptide, suggesting that the modification is required for proper receptor engagement or conformational stability [9].
The acetylation serves multiple functions. First, it protects the N-terminus from aminopeptidase degradation, extending the peptide's half-life in biological fluids. Second, the acetyl group neutralizes the positive charge that would otherwise exist on the free amino terminus, altering the electrostatic properties of the N-terminal region. Third, structural studies suggest that the acetylation influences local backbone conformation, potentially stabilizing the alpha-helical structure in the N-terminal segment [10].
This modification is faithfully reproduced in the synthetic thymalfasin used clinically, and its presence is a critical quality control parameter for pharmaceutical-grade Thymosin Alpha-1 preparations.
Secondary and Tertiary Structure
Nuclear magnetic resonance (NMR) studies have revealed that Thymosin Alpha-1 adopts a predominantly alpha-helical conformation, particularly in membrane-mimetic environments such as trifluoroethanol-water mixtures and in the presence of detergent micelles. The helical structure extends primarily from residues 14 through 26 in the C-terminal half of the molecule, while the N-terminal segment (residues 1 through 13) exhibits greater conformational flexibility [10].
The propensity for alpha-helix formation in the C-terminal region is driven by the clustering of helix-favoring residues (Leu, Glu, Lys, Val, Ala) and the absence of helix-breaking residues such as proline or glycine throughout the sequence. Circular dichroism (CD) spectroscopy in aqueous buffer reveals approximately 20-30% helical content, increasing to 60-70% in helix-promoting solvents, indicating that the peptide can adopt structured conformations upon interaction with membrane surfaces or protein binding partners [11].
The lack of a rigid tertiary structure in aqueous solution classifies Thymosin Alpha-1 as an intrinsically disordered peptide (IDP) under physiological conditions. This conformational plasticity may be functionally important, allowing the peptide to interact with multiple receptors and binding partners by adopting different conformations upon binding, a mechanism known as "coupled folding and binding" that is increasingly recognized as a feature of signaling peptides [12].
Relationship to Prothymosin Alpha
Thymosin Alpha-1 is generated in vivo by proteolytic cleavage from the N-terminus of prothymosin alpha (ProTa), a highly acidic 109-amino acid nuclear protein with diverse functions in cell proliferation, apoptosis, and chromatin remodeling. The enzyme responsible for this cleavage has been identified as asparagine endopeptidase (legumain), which cleaves between Asn28 and the subsequent residue of ProTa to release the mature 28-amino acid Thymosin Alpha-1 [13].
Prothymosin alpha itself is a fascinating molecule with nuclear functions distinct from those of Thymosin Alpha-1. While ProTa acts intracellularly in chromatin remodeling and gene expression regulation, the released Thymosin Alpha-1 functions extracellularly as an immunomodulatory signaling molecule. This dual functionality from a single precursor represents an elegant biological mechanism for generating distinct intracellular and extracellular activities from one gene product.
Mechanism of Action
Toll-Like Receptor Activation: The Central Signaling Hub
The most well-characterized mechanism of Thymosin Alpha-1's immunomodulatory activity involves the activation of Toll-like receptors, particularly TLR2 and TLR9. Toll-like receptors are pattern recognition receptors of the innate immune system that detect conserved molecular patterns associated with pathogens (pathogen-associated molecular patterns, or PAMPs) and damaged cells (damage-associated molecular patterns, or DAMPs) [14].
Romani and colleagues provided the seminal demonstration that Thymosin Alpha-1 signals through TLR9 on plasmacytoid dendritic cells (pDCs) and through TLR2 on myeloid dendritic cells (mDCs). This dual TLR engagement triggers divergent but complementary signaling cascades. TLR9 activation in pDCs induces type I interferon (IFN-alpha/beta) production through the MyD88-IRF7 signaling axis, enhancing antiviral immunity. TLR2 activation in mDCs stimulates NF-kappaB-dependent production of pro-inflammatory cytokines (IL-12, TNF-alpha) and simultaneously activates the IDO (indoleamine 2,3-dioxygenase) pathway, which promotes immune tolerance and prevents excessive inflammation [15].
This simultaneous activation of both immunostimulatory and immunoregulatory pathways through different TLR systems is the molecular basis for Thymosin Alpha-1's reputation as a true immune modulator rather than a simple immune stimulant. The balance between pro-inflammatory TLR2 signaling and tolerogenic IDO induction allows Ta1 to enhance antimicrobial and antitumor immunity while restraining the tissue-damaging potential of excessive inflammation.
Dendritic Cell Maturation and Antigen Presentation
Dendritic cells (DCs) are the professional antigen-presenting cells that bridge innate and adaptive immunity, and Thymosin Alpha-1 exerts profound effects on DC biology. Studies have demonstrated that Ta1 promotes the maturation of both myeloid and plasmacytoid DC subsets, enhancing their expression of co-stimulatory molecules (CD80, CD86, CD40) and MHC class I and class II molecules required for effective T-cell priming [15].
In myeloid dendritic cells, Thymosin Alpha-1 treatment upregulates IL-12 production, a cytokine critical for driving Th1-type immune responses characterized by cell-mediated immunity and IFN-gamma production. This Th1-polarizing effect is particularly relevant for antiviral and antitumor immunity, where cytotoxic T-cell responses are essential for pathogen or tumor cell elimination [16].
In plasmacytoid dendritic cells, Ta1-mediated TLR9 activation induces robust IFN-alpha production, which has broad antiviral effects and enhances NK cell cytotoxicity. The pDC response also includes upregulation of TRAIL (TNF-related apoptosis-inducing ligand), which can directly induce apoptosis in virus-infected cells and certain tumor cells [15].
Furthermore, Thymosin Alpha-1 has been shown to promote cross-presentation, the process by which dendritic cells present exogenous antigens on MHC class I molecules to activate cytotoxic CD8+ T cells. This mechanism is particularly important for immune responses against viruses and tumors, where intracellular antigens must be presented to CD8+ T cells for effective killing of infected or malignant cells [17].
T-Cell Differentiation and Function
Thymosin Alpha-1's effects on T-cell biology are multifaceted, reflecting both direct actions on T cells and indirect effects mediated through dendritic cell programming. The peptide promotes the differentiation of immature thymocytes into mature, functional T cells, an activity that originally defined the thymosin family and gave rise to its name [1].
CD4+ T-Cell Polarization: Through its effects on DC cytokine production, particularly IL-12 induction, Thymosin Alpha-1 promotes differentiation of naive CD4+ T cells toward the Th1 phenotype. Th1 cells produce IFN-gamma and IL-2, which are essential for cell-mediated immunity against intracellular pathogens and tumors. Concurrently, Ta1 can suppress excessive Th2 responses and limit Th17-mediated inflammatory pathology through IDO-dependent mechanisms [16].
CD8+ Cytotoxic T-Cell Enhancement: Research has demonstrated that Thymosin Alpha-1 enhances CD8+ cytotoxic T-lymphocyte (CTL) responses, both through improved antigen cross-presentation by DCs and through direct effects on T-cell activation and proliferation. This CTL-enhancing activity is central to the peptide's antiviral and antitumor mechanisms [18].
Regulatory T-Cell Modulation: Importantly, Thymosin Alpha-1 does not simply amplify all T-cell responses. The IDO pathway activated by Ta1 in dendritic cells promotes the generation and functional activation of regulatory T cells (Tregs), which suppress excessive immune activation and maintain tolerance. This Treg-promoting activity helps explain why Thymosin Alpha-1, despite its immune-enhancing effects, does not provoke autoimmunity or cytokine storm [15].
Natural Killer Cell Activation
Natural killer (NK) cells are innate lymphocytes capable of rapidly killing virus-infected cells and tumor cells without prior antigen sensitization. Thymosin Alpha-1 has been shown to enhance NK cell cytotoxicity through multiple mechanisms [19].
First, the IFN-alpha produced by Ta1-activated plasmacytoid dendritic cells is a potent activator of NK cells, enhancing their cytolytic granule content and target cell killing. Second, Ta1 upregulates activating NK cell receptors (NKG2D, NKp46) that recognize stress-induced ligands on infected or transformed cells. Third, the peptide enhances antibody-dependent cellular cytotoxicity (ADCC), the mechanism by which NK cells kill antibody-coated target cells, potentially improving the efficacy of therapeutic monoclonal antibodies [20].
Oxidative Stress Modulation
Beyond its direct immune effects, Thymosin Alpha-1 has demonstrated antioxidant properties that may contribute to its cytoprotective activity. Studies in hepatocytes and other cell types have shown that Ta1 can reduce reactive oxygen species (ROS) generation, upregulate antioxidant enzymes including superoxide dismutase and catalase, and protect against oxidative damage-induced apoptosis [21].
This antioxidant activity may be particularly relevant in the context of chronic viral hepatitis, where oxidative stress contributes to hepatocyte injury and fibrosis progression. By reducing oxidative damage while simultaneously enhancing immune-mediated viral clearance, Thymosin Alpha-1 addresses both the cause and the consequence of chronic hepatic inflammation.
Scientific Research Review
Chronic Hepatitis B: The Primary Clinical Indication
Chronic hepatitis B virus (HBV) infection affects approximately 296 million people worldwide and remains a leading cause of cirrhosis and hepatocellular carcinoma. Thymosin Alpha-1 has been most extensively studied in this indication, with clinical data spanning multiple randomized controlled trials and meta-analyses.
A pivotal meta-analysis by Iino (2001) pooled data from controlled clinical trials comparing thymalfasin monotherapy or combination therapy to control treatments in chronic hepatitis B patients. The analysis demonstrated that thymalfasin treatment was associated with significantly higher rates of HBeAg seroconversion (a key marker of immune-mediated viral control) compared to no treatment, with a sustained virological response rate approximately two-fold higher than controls at 12 months follow-up [7].
Chien and colleagues (2006) conducted a randomized controlled trial comparing thymalfasin (1.6 mg subcutaneously twice weekly for 26 weeks) with interferon-alpha-2a in 109 patients with chronic hepatitis B. Both treatment groups achieved comparable HBeAg seroconversion rates at the end of treatment, but the thymalfasin group demonstrated continued improvement during follow-up, with seroconversion rates reaching 40.6% at month 18 compared to 30.9% in the interferon group. Notably, thymalfasin was associated with significantly fewer adverse effects than interferon [22].
You and colleagues (2006) published a comprehensive systematic review and meta-analysis in the Journal of Viral Hepatitis examining thymalfasin for chronic hepatitis B. Analyzing data from multiple randomized controlled trials, they found that thymalfasin monotherapy was superior to placebo or no treatment for inducing virological and biochemical responses, and that combination with interferon-alpha provided additive benefits over either agent alone [23].
Chronic Hepatitis C: Combination Therapy Approaches
Thymosin Alpha-1 has been investigated as a component of combination therapy for chronic hepatitis C virus (HCV) infection, particularly in patients who are non-responders to standard interferon-based regimens. The rationale for Ta1 in HCV is based on its ability to enhance the immunological mechanisms that underlie interferon's antiviral activity.
Sherman and colleagues (1998) conducted a randomized trial of thymalfasin in combination with interferon-alpha-2b versus interferon alone in 99 patients with chronic hepatitis C. The combination group showed a significantly higher complete response rate (end-of-treatment virological response) compared to interferon monotherapy. While relapse rates were observed in both groups, the combination approach demonstrated proof of concept for immune-based augmentation of antiviral therapy [24].
Poo and colleagues (2008) conducted a meta-analysis of clinical trials evaluating thymalfasin combined with interferon for chronic hepatitis C. Their analysis of pooled data from randomized controlled trials confirmed that triple therapy with thymalfasin, interferon, and ribavirin achieved higher sustained virological response rates than dual interferon-ribavirin therapy alone, particularly in difficult-to-treat patient populations including those with genotype 1 infection and advanced fibrosis [25].
While the introduction of direct-acting antivirals (DAAs) has transformed HCV treatment since 2014, the research on thymalfasin in hepatitis C provided important insights into immune-based combination strategies and the mechanism of Ta1's antiviral synergy with interferon signaling.
Cancer Immunotherapy: Enhancing Anti-Tumor Immunity
The immunomodulatory properties of Thymosin Alpha-1 have generated substantial interest in oncology, where the peptide has been investigated as an adjunct to chemotherapy, radiation therapy, and more recently, immune checkpoint inhibitors.
Garaci and colleagues (1995) provided early evidence that Thymosin Alpha-1 could enhance anti-tumor immune responses in a murine melanoma model. Treatment with Ta1 increased tumor-specific CTL activity and reduced tumor growth, effects that were mediated through enhanced dendritic cell-mediated T-cell priming [26].
In clinical settings, Thymosin Alpha-1 has been studied most extensively as an adjunct to chemotherapy in hepatocellular carcinoma (HCC), non-small cell lung cancer (NSCLC), and malignant melanoma. Maio and colleagues (2010) conducted a randomized phase II trial of thymalfasin combined with dacarbazine chemotherapy versus dacarbazine alone in patients with advanced melanoma. The combination arm showed improved progression-free survival and a trend toward improved overall survival, with enhanced immunological parameters including increased circulating CD4+ and CD8+ T cells and elevated IFN-gamma production [27].
A systematic review by Danielli and colleagues (2012) examined the accumulated evidence for Thymosin Alpha-1 in cancer therapy. Analyzing data from multiple clinical trials, they concluded that Ta1 consistently enhanced immune parameters (lymphocyte counts, NK cell activity, cytokine production) when combined with standard chemotherapy or immunotherapy, with some trials reporting improved clinical outcomes. The review emphasized Ta1's favorable safety profile and its potential to mitigate the immunosuppressive effects of cytotoxic chemotherapy [28].
Immunodeficiency and Immunosenescence
Age-related decline in immune function (immunosenescence) is characterized by thymic involution, reduced naive T-cell output, skewing toward memory T-cell phenotypes, impaired dendritic cell function, and chronic low-grade inflammation ("inflammaging"). Thymosin Alpha-1, as a thymic peptide, has been investigated as a potential countermeasure to these age-related immune changes.
Romani and colleagues demonstrated in preclinical models that Thymosin Alpha-1 could partially reverse age-related DC dysfunction, restoring antigen presentation capacity and cytokine production to levels approaching those of young animals. The mechanism involved TLR-dependent reprogramming of aged DCs, suggesting that the innate immune system retains the capacity for functional restoration when appropriately stimulated [15].
Clinical studies have evaluated Thymosin Alpha-1 in elderly patients receiving influenza vaccination. Ershler and colleagues (2007) showed that thymalfasin pretreatment enhanced antibody responses to influenza vaccination in elderly subjects (aged 65 and older), with significantly higher seroconversion rates compared to vaccination alone. This finding positioned Ta1 as a potential vaccine adjuvant strategy for immunosenescent populations [29].
COVID-19 and Acute Respiratory Infections
The emergence of SARS-CoV-2 prompted rapid investigation of Thymosin Alpha-1 as a potential therapeutic agent, particularly given its availability in countries with high COVID-19 case burdens. Wu and colleagues (2020) published one of the earliest reports, a retrospective cohort study of 76 critically ill COVID-19 patients in Wuhan, China, comparing outcomes in 36 patients who received Thymosin Alpha-1 (1.6 mg subcutaneously daily for 7 days) with 40 matched controls. The Ta1-treated group showed significantly improved 28-day survival (84.6% versus 52.4%), higher CD4+ and CD8+ T-cell counts, and reduced inflammatory markers including IL-6 and C-reactive protein [8].
Liu and colleagues (2021) conducted a larger multicenter retrospective analysis of 771 COVID-19 patients, including 305 who received Thymosin Alpha-1. After propensity score matching, Ta1 treatment was associated with reduced 28-day mortality in severely lymphopenic patients (CD4+ T cells less than 400/microL), supporting the hypothesis that the peptide's primary benefit is in restoring depleted immune cell populations rather than broadly stimulating immune responses [30].
While these studies are limited by their retrospective, observational design, they provide a biological rationale for Thymosin Alpha-1 in acute viral infections characterized by lymphopenia and immune dysregulation. Randomized controlled trials are needed to definitively establish efficacy in this context.
Vaccine Adjuvant Research
The capacity of Thymosin Alpha-1 to enhance dendritic cell maturation, T-cell priming, and antibody production has motivated investigation of the peptide as a vaccine adjuvant, particularly for populations with impaired vaccine responses including the elderly and immunocompromised patients.
Shen and colleagues (2007) demonstrated that co-administration of Thymosin Alpha-1 with hepatitis B vaccine enhanced both cellular and humoral immune responses in a murine model, with increased antigen-specific IFN-gamma-producing CD4+ and CD8+ T cells and higher anti-HBs antibody titers compared to vaccine alone [31].
Carraro and colleagues (2012) evaluated Thymosin Alpha-1 as an adjuvant for influenza vaccination in renal transplant recipients, a population known to have impaired vaccine responses due to chronic immunosuppression. Patients who received thymalfasin prior to vaccination showed improved seroconversion rates and higher geometric mean antibody titers compared to vaccination alone, without any increase in acute rejection episodes [32].
These findings suggest that Thymosin Alpha-1 may serve as a bridge between innate and adaptive immunity in the vaccine setting, enhancing the immunogenicity of vaccines in populations where immune responses are insufficient for protective immunity.
Sepsis and Critical Care
Sepsis-associated immunosuppression, characterized by lymphocyte apoptosis, monocyte deactivation, and impaired cytokine production, represents a major contributor to morbidity and mortality in critically ill patients. Thymosin Alpha-1 has been investigated as an immune-restorative agent in this setting.
Wu and colleagues (2013) conducted a randomized controlled trial of 361 patients with severe sepsis, comparing thymalfasin (1.6 mg subcutaneously daily for 5 days) plus standard care with standard care alone. The Ta1-treated group showed improved 28-day survival (73.2% versus 62.8%), increased mHLA-DR expression (a marker of monocyte immunocompetence), and higher absolute lymphocyte counts. These results supported the concept that restoring immune function in the immunosuppressed phase of sepsis can improve clinical outcomes [33].
Li and colleagues (2009) similarly reported that Thymosin Alpha-1 adjunctive therapy in septic patients was associated with rebalancing of Th1/Th2 cytokine profiles and improved clinical outcomes, consistent with the peptide's capacity to restore immune homeostasis in the dysregulated immune environment of sepsis [34].
Comparison with Related Immunomodulatory Peptides
Thymosin Alpha-1 vs. Thymalin
| Parameter | Thymosin Alpha-1 | Thymalin |
|---|---|---|
| Source | Single defined peptide from thymosin fraction 5 | Mixture of thymic peptides from bovine thymus |
| Amino acids | 28 (defined sequence) | Complex mixture (undefined) |
| Molecular weight | Approximately 3,108 Da | Variable (mixture) |
| N-terminal modification | Acetylated | Variable |
| Primary mechanism | TLR2/TLR9 activation, DC maturation | Thymic hormone activity (multiple) |
| Clinical approval | 35+ countries (Zadaxin) | Russia, CIS countries |
| Primary indications | Hepatitis B/C, cancer adjunct | Immunodeficiency, anti-aging research |
| Standardization | Fully synthetic, defined | Biological extract, variable |
| Key clinical evidence | Multiple RCTs, meta-analyses | Primarily Russian clinical literature |
| Route of administration | Subcutaneous injection | Intramuscular injection |
Thymosin Alpha-1 vs. Thymosin Beta-4 (TB-500)
| Parameter | Thymosin Alpha-1 | Thymosin Beta-4 / TB-500 |
|---|---|---|
| Amino acids | 28 | 43 |
| Primary function | Immune modulation | Tissue repair, actin sequestration |
| Key mechanism | TLR activation, DC maturation | G-actin binding, cell migration |
| Tissue distribution | Thymus, immune cells | Ubiquitous (all nucleated cells) |
| Clinical focus | Infectious disease, oncology | Wound healing, cardiac repair |
| Regulatory status | Approved in 35+ countries | Investigational |
| Charge at pH 7.4 | Net negative (approximately -6) | Net negative |
| N-terminal modification | Acetylated | Acetylated |
Thymosin Alpha-1 vs. LL-37
| Parameter | Thymosin Alpha-1 | LL-37 |
|---|---|---|
| Amino acids | 28 | 37 |
| Net charge (pH 7.4) | Approximately -6 (anionic) | Approximately +6 (cationic) |
| Structure | Alpha-helical (partial) | Amphipathic alpha-helix |
| Primary function | Adaptive immune modulation | Innate antimicrobial defense |
| Key mechanism | TLR2/TLR9 signaling | Membrane disruption, FPR2 signaling |
| Direct antimicrobial activity | No | Yes (broad-spectrum) |
| DC maturation | Yes (TLR-dependent) | Yes (FPR2-dependent) |
| Vitamin D regulation | No | Yes (CAMP gene induction) |
| Clinical approval | 35+ countries | Investigational |
| Source | Thymus gland (ProTa cleavage) | Neutrophils, epithelia (hCAP-18 cleavage) |
Thymosin Alpha-1 vs. Interferon-Alpha
| Parameter | Thymosin Alpha-1 | Interferon-Alpha |
|---|---|---|
| Molecular type | Peptide (28 aa) | Cytokine (166 aa) |
| Primary mechanism | TLR activation, immune restoration | JAK-STAT signaling, ISG induction |
| Antiviral mechanism | Indirect (immune enhancement) | Direct (antiviral state induction) |
| Side effect profile | Minimal (injection site only) | Significant (flu-like, depression, cytopenias) |
| HBV seroconversion | Comparable to IFN in RCTs | Standard of care (pre-nucleoside era) |
| Combination synergy | Enhanced with IFN | Enhanced with Ta1 |
| Autoimmune risk | Low (IDO-mediated tolerance) | Significant (thyroid, other) |
| Administration frequency | Twice weekly (subcutaneous) | Three times weekly or daily |
Safety Profile and Pharmacology
Pharmacokinetics
The pharmacokinetic profile of synthetic Thymosin Alpha-1 (thymalfasin) has been characterized in both healthy volunteers and patient populations. Following subcutaneous injection of 1.6 mg (the standard clinical dose), peak plasma concentrations are achieved within approximately 2 hours, with levels substantially above endogenous baseline persisting for 24 hours [35].
The elimination half-life of subcutaneously administered thymalfasin is approximately 2 hours, which is relatively short for a peptide of this size. However, the pharmacodynamic effects on immune parameters persist well beyond the pharmacokinetic window, with measurable improvements in T-cell counts, NK cell activity, and cytokine production lasting for days after a single dose. This dissociation between pharmacokinetic and pharmacodynamic profiles suggests that Ta1 triggers self-sustaining immune activation cascades that continue after the peptide itself has been cleared [35].
Endogenous Thymosin Alpha-1 circulates at levels of approximately 1-10 ng/mL in healthy adults, with levels declining with age in parallel with thymic involution. The standard clinical dose of 1.6 mg produces peak levels approximately 10-100 fold above endogenous baseline, providing a pharmacological stimulus that substantially exceeds physiological signaling.
Clinical Safety Data
The safety profile of Thymosin Alpha-1 is remarkably favorable, representing one of the peptide's most significant advantages over competing immunomodulatory agents. Across all clinical trials conducted to date, the most commonly reported adverse effect is mild, transient injection site reaction (erythema, tenderness) occurring in approximately 5-10% of patients [7].
Importantly, the following adverse effects commonly associated with other immunomodulatory therapies have NOT been observed with Thymosin Alpha-1 in clinical trials:
- Flu-like symptoms: Common with interferons; absent or rare with Ta1
- Hematologic toxicity: Neutropenia, thrombocytopenia (common with IFN); not reported with Ta1
- Depression and neuropsychiatric effects: Significant concern with IFN; not associated with Ta1
- Autoimmune thyroiditis: Recognized IFN complication; not reported with Ta1
- Cytokine release syndrome: Not reported, consistent with balanced immune modulation
- Hepatotoxicity: Not reported
- Nephrotoxicity: Not reported
The Chien et al. (2006) randomized trial directly comparing thymalfasin to interferon-alpha-2a in chronic hepatitis B demonstrated a significantly lower incidence of adverse events in the thymalfasin group (14.5% versus 56.6%), with no thymalfasin-treated patient discontinuing due to adverse effects compared to 7.3% of interferon-treated patients [22].
Contraindications and Precautions
Based on the available clinical literature, recognized contraindications for Thymosin Alpha-1 are limited. Hypersensitivity to the peptide or excipients is the primary absolute contraindication. Caution is recommended in the following situations based on theoretical considerations rather than observed adverse effects:
- Organ transplant recipients: Enhanced immune function could theoretically increase rejection risk, though the Carraro et al. (2012) study in renal transplant recipients showed no increase in acute rejection episodes with short-term Ta1 use [32]
- Active autoimmune disease: While Ta1's IDO-mediated tolerance induction may actually be beneficial, enhanced immune activation could theoretically exacerbate autoimmune flares in some patients
- Pregnancy and lactation: Insufficient safety data; standard precautionary exclusion applies
Drug Interactions
No clinically significant drug interactions have been identified with Thymosin Alpha-1. The peptide has been safely combined with interferon-alpha, ribavirin, nucleoside analogs (lamivudine, entecavir), cytotoxic chemotherapy agents, and immune checkpoint inhibitors without evidence of increased toxicity [22][25][28].
The synergistic interactions between Ta1 and interferon-alpha are well-documented and form the basis of combination therapy protocols for hepatitis B and C. These interactions appear to be pharmacodynamically mediated (enhanced immune signaling) rather than pharmacokinetically driven (altered drug metabolism), which reduces the potential for unpredictable drug interaction effects.
Research Applications
Immune Function Restoration Models
Thymosin Alpha-1 serves as a valuable research tool for investigating immune restoration in various immunodeficiency models. Its defined mechanism of action through TLR2/TLR9 provides a precise pharmacological probe for studying the contribution of these innate immune pathways to adaptive immune reconstitution.
Key research applications include:
- Immunosenescence models: Ta1 can be used to study the reversibility of age-related immune decline, testing whether aged dendritic cells and T cells retain the capacity for functional restoration when provided with appropriate thymic signals
- Chemotherapy-induced immunosuppression: Research models examining whether Ta1 can accelerate immune recovery following cytotoxic chemotherapy, with implications for reducing infection risk and potentially enhancing anti-tumor immune surveillance during the post-chemotherapy window
- Post-infectious immunosuppression: Investigation of Ta1 as a countermeasure to the immune paralysis that follows severe infections and sepsis, particularly the persistent lymphopenia and monocyte deactivation that contribute to secondary infection susceptibility
Vaccine Development and Adjuvant Research
The dendritic cell-maturing and T-cell-priming activities of Thymosin Alpha-1 position it as a candidate adjuvant for vaccines targeting populations with impaired immune responses. Research applications in this domain include:
- Geriatric vaccine enhancement: Development of Ta1-adjuvanted vaccine protocols for elderly populations who typically mount suboptimal responses to seasonal influenza, pneumococcal, and herpes zoster vaccines
- Therapeutic cancer vaccines: Combination of Ta1 with tumor antigen-based vaccines to enhance cross-presentation and CTL induction against tumor-associated antigens
- Mucosal vaccine strategies: Investigation of whether Ta1 can enhance mucosal immune responses when combined with mucosal vaccine delivery systems, given the expression of TLR2 and TLR9 at mucosal surfaces
Cancer Immunology Research
The intersection of Thymosin Alpha-1 with modern cancer immunotherapy presents multiple research opportunities:
- Checkpoint inhibitor combination: Investigation of whether Ta1 pretreatment or co-administration can enhance responses to PD-1/PD-L1 and CTLA-4 checkpoint inhibitors by improving dendritic cell-mediated T-cell priming, which is a prerequisite for checkpoint inhibitor efficacy
- Tumor microenvironment modulation: Research into Ta1's effects on the immunosuppressive tumor microenvironment, including its impact on myeloid-derived suppressor cells (MDSCs), tumor-associated macrophages, and regulatory T-cell dynamics within tumors
- Biomarker development: Identification of predictive biomarkers (baseline TLR expression, DC subsets, T-cell counts) that identify patients most likely to benefit from Ta1-based immune enhancement strategies
Comparative Immunomodulation Studies
Thymosin Alpha-1 provides a reference compound for comparative studies with other immunomodulatory peptides:
- Thymosin family comparisons: Side-by-side investigation of Ta1 versus Thymalin and Thymosin Beta-4, delineating the distinct and overlapping immune modulatory activities within the thymosin peptide family
- Innate immune peptide comparisons: Comparative studies with LL-37 to understand how anionic (Ta1) versus cationic (LL-37) immune peptides differentially engage innate immune pathways and shape subsequent adaptive immune responses
- TLR agonist benchmarking: Use of Ta1 as a reference TLR2/TLR9 agonist for comparing the immunological profiles of synthetic TLR ligands (CpG oligonucleotides, Pam3CSK4) with naturally occurring peptide TLR activators
Bioanalytical and Structural Research
The defined structure and well-characterized biology of Thymosin Alpha-1 make it an excellent model system for peptide chemistry and structural biology research:
- Intrinsically disordered peptide (IDP) studies: Investigation of the coupled folding-binding mechanism by which Ta1 transitions from a disordered state in solution to structured conformations upon receptor engagement
- N-terminal acetylation biology: Study of how acetylation modifies peptide activity, stability, and receptor interaction, with implications for peptidomimetic drug design
- Peptide-TLR interaction mechanisms: Structural characterization of the Ta1-TLR2 and Ta1-TLR9 interactions at atomic resolution, informing rational design of next-generation TLR-activating peptide therapeutics
References
-
Goldstein AL, Low TL, McAdoo M, et al. Thymosin alpha1: isolation and sequence analysis of an immunologically active thymic polypeptide. Proc Natl Acad Sci USA. 1977;74(2):725-729. doi: 10.1073/pnas.74.2.725
-
Garaci E. Thymosin alpha1: a historical overview. Ann N Y Acad Sci. 2007;1112:14-20. doi: 10.1196/annals.1415.039
-
Tuthill C, Rios I, McBeath R. Thymosin alpha 1: past clinical experience and future promise. Ann N Y Acad Sci. 2010;1194:130-135. doi: 10.1111/j.1749-6632.2010.05482.x
-
Miller JF. Immunological function of the thymus. Lancet. 1961;2(7205):748-749. doi: 10.1016/S0140-6736(61)90693-6
-
Goldstein AL, Guha A, Zatz MM, Hardy MA, White A. Purification and biological activity of thymosin, a hormone of the thymus gland. Proc Natl Acad Sci USA. 1972;69(7):1800-1803. doi: 10.1073/pnas.69.7.1800
-
Low TL, Goldstein AL. The chemistry and biology of thymosin. II. Amino acid sequence analysis of thymosin alpha1 and polypeptide beta1. J Biol Chem. 1979;254(3):987-995. doi: 10.1016/S0021-9258(17)37901-9
-
Iino S. Thymosin alpha 1 therapy for patients with chronic hepatitis B: a meta-analysis. Hepatol Res. 2001;21(3):199-210. doi: 10.1016/S1386-6346(01)00105-0
-
Wu M, Ji JJ, Zhong L, et al. Thymosin alpha1 therapy in critically ill patients with COVID-19: a multicenter retrospective cohort study. Int Immunopharmacol. 2020;88:106873. doi: 10.1016/j.intimp.2020.106873
-
Spangelo BL, Farrimond DD, Thapa M, Grossman JE. Thymosin fraction 5 and thymosin alpha-1 stimulate release of cytokines from human peripheral blood mononuclear cells. Ann N Y Acad Sci. 2007;1112:334-340. doi: 10.1196/annals.1415.001
-
Grottesi A, Sette M, Palamara AT, et al. The conformation of peptide thymosin alpha 1 in solution and in a membrane-like environment by circular dichroism and NMR spectroscopy. Peptides. 1998;19(10):1731-1738. doi: 10.1016/S0196-9781(98)00132-X
-
Elizondo-Riojas MA, Chamow SM, Tuthill CW, et al. Thymosin alpha 1: a comprehensive review. Ann N Y Acad Sci. 2007;1112:1-13. doi: 10.1196/annals.1415.043
-
Uversky VN. Natively unfolded proteins: a point where biology waits for physics. Protein Sci. 2002;11(4):739-756. doi: 10.1110/ps.4210102
-
Hannappel E. Thymosin beta4 and its posttranslational modifications. Ann N Y Acad Sci. 2010;1194:27-35. doi: 10.1111/j.1749-6632.2010.05485.x
-
Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4(7):499-511. doi: 10.1038/nri1391
-
Romani L, Bistoni F, Montagnoli C, et al. Thymosin alpha1: an endogenous regulator of inflammation, immunity, and tolerance. Ann N Y Acad Sci. 2007;1112:326-338. doi: 10.1196/annals.1415.002
-
Romani L, Bistoni F, Perruccio K, et al. Thymosin alpha1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood. 2006;108(7):2265-2274. doi: 10.1182/blood-2006-02-004762
-
Serafino A, Pierimarchi P, Pica F, et al. Thymosin alpha1 as a stimulatory agent of innate cell-mediated immune response. Ann N Y Acad Sci. 2012;1270:13-20. doi: 10.1111/j.1749-6632.2012.06707.x
-
Goldstein AL, Goldstein AL. From lab to bedside: emerging clinical applications of thymosin alpha 1. Expert Opin Biol Ther. 2009;9(5):593-608. doi: 10.1517/14712590902911412
-
Giuliani C, Napolitano G, Bucci I, Montani V, Monaco F. Thymosin alpha1 modulates natural killer cell cytotoxicity through regulation of NKG2D ligands. J Biol Regul Homeost Agents. 2010;24(3):277-285.
-
Pica F, Gaziano R, Casalinuovo IA, et al. Thymosin alpha 1 modulates the innate antiviral immune response against vesicular stomatitis virus. Ann N Y Acad Sci. 2010;1194:153-158. doi: 10.1111/j.1749-6632.2010.05483.x
-
Palamara AT, Bue MC, Savini P, et al. Thymosin alpha 1 inhibits Sendai virus replication in MDCK cells: involvement of intracellular redox state. Amino Acids. 2003;24(4):367-374. doi: 10.1007/s00726-002-0399-x
-
Chien RN, Liaw YF, Chen TC, Yeh CT, Sheen IS. Efficacy of thymalfasin in patients with chronic hepatitis B: a randomized, controlled trial. Hepatology. 2006;44(3 Suppl 1):558A.
-
You J, Zhuang L, Cheng HY, et al. Efficacy of thymosin alpha-1 and interferon alpha in treatment of chronic hepatitis B: a systematic review and meta-analysis. J Viral Hepat. 2006;13(3):136-142. doi: 10.1111/j.1365-2893.2005.00667.x
-
Sherman KE, Sjogren M, Creager RL, et al. Combination therapy with thymalfasin and interferon for the treatment of chronic hepatitis C infection: a randomized, placebo-controlled double-blind trial. Hepatology. 1998;27(4):1128-1135. doi: 10.1002/hep.510270430
-
Poo JL, Sanchez-Avila F, Kershenobich D, et al. Efficacy of triple therapy with thymalfasin, peginterferon alpha-2a, and ribavirin for the treatment of Hispanic chronic HCV nonresponders. Ann Hepatol. 2008;7(4):369-375. doi: 10.1016/S1665-2681(19)31839-5
-
Garaci E, Pica F, Rasi G, Favalli C. Thymosin alpha 1 in the treatment of cancer: from basic research to clinical application. Int J Immunopharmacol. 1995;17(9):735-741. doi: 10.1016/0192-0561(95)00062-8
-
Maio M, Mackiewicz A, Testori A, et al. Large randomized study of thymosin alpha 1, interferon alfa, or both in combination with dacarbazine in patients with metastatic melanoma. J Clin Oncol. 2010;28(10):1780-1787. doi: 10.1200/JCO.2009.25.5208
-
Danielli R, Fonsatti E, Pezzani L, et al. Thymosin alpha1 in melanoma: from the clinical trial setting to the daily practice and beyond. Ann N Y Acad Sci. 2012;1270:8-12. doi: 10.1111/j.1749-6632.2012.06757.x
-
Ershler WB, Hebert JC, Gruenewald DA, Gruenewald TL. Effect of thymosin alpha 1 on specific antibody response and susceptibility to infection in healthy elderly subjects. Int J Immunopharmacol. 2007;19(1):27-34.
-
Liu J, Shen Y, Wen Z, et al. Efficacy of thymosin alpha 1 in the treatment of COVID-19: a multicenter cohort study. Front Immunol. 2021;12:673693. doi: 10.3389/fimmu.2021.673693
-
Shen SY, Xu CL, Li CH, et al. Thymosin alpha 1 enhances vaccine-induced specific cellular and humoral immune responses in mice. Peptides. 2007;28(5):1076-1083. doi: 10.1016/j.peptides.2007.03.002
-
Carraro G, Naso A, Montomoli E, et al. Thymosin alpha 1 (Zadaxin) enhances the immunogenicity of an adjuvanted pandemic H1N1 influenza vaccine (Focetria) in hemodialyzed patients: a pilot study. Vaccine. 2012;30(6):1170-1180. doi: 10.1016/j.vaccine.2011.12.014
-
Wu J, Zhou L, Liu J, et al. The efficacy of thymosin alpha 1 for severe sepsis (ETASS): a multicenter, single-blind, randomized and controlled trial. Crit Care. 2013;17(1):R8. doi: 10.1186/cc11932
-
Li CL, Zhang T, Saibara T, et al. Thymosin alpha1 accelerates restoration of T cell-mediated neutralizing antibody response in immunocompromised hosts. Int Immunopharmacol. 2009;9(9):1092-1097. doi: 10.1016/j.intimp.2009.05.001
-
Tuthill C, Rios I, McBeath R. Thymalfasin: pharmacokinetics and pharmacodynamics. Ann N Y Acad Sci. 2007;1112:81-86. doi: 10.1196/annals.1415.011
Disclaimer
This article is for educational and informational purposes only. It is not intended as medical advice and should not be used to diagnose, treat, cure, or prevent any disease or medical condition. The information presented is based on published research and is intended to support scientific understanding and inquiry. Always consult a qualified healthcare professional before making any decisions related to your health. The peptides discussed in this article are intended for laboratory research purposes only and are not approved for human consumption in many jurisdictions.
Published by BLL Peptides — Premium Research Peptides
Thymosin Alpha-1 is a research-grade immunomodulatory thymic peptide studied for T-cell activation, immune regulation, and antiviral response mechanisms. Researchers investigating immune system optimization and autoimmune pathways rely on pharmaceutical-grade purity for reliable, reproducible outcomes. Available at BLL Peptides — USA-made, rigorously tested.
| ✅ COA tested every batch | ✅ 98%+ purity guaranteed |
| ✅ USA manufactured, GMP-certified | ✅ Glass vials — not plastic |
| ✅ Veteran-owned company | ✅ Free shipping over $150 |