CJC-1295 With DAC 5mg (3ml)
CJC-1295 with DAC is a research-grade extended-release GHRH analog using Drug Affinity Complex technology, studied for prolonged half-life, sustained GH and IGF-1 elevation, and body composition research. Researchers investigating long-duration growth hormone stimulation rely on pharmaceutical-grade purity for valid outcomes. Available at BLL Peptides — USA-made, rigorously tested. ✅ COA tested every batch✅ 98%+ purity…
Description
CJC-1295 With DAC: Complete Research Guide – Long-Acting GHRH Analog Mechanisms, Sustained GH Stimulation, and Research Applications
Last updated: March 2026
Executive Summary
CJC-1295 with DAC (Drug Affinity Complex) is a long-acting synthetic analog of growth hormone-releasing hormone (GHRH) developed by ConjuChem Biotechnologies (Montreal, Canada). It consists of a modified 30-amino acid GHRH peptide covalently linked to a reactive chemical moiety — maleimidopropionic acid (MPA) — at the C-terminal lysine residue (Lys30). This MPA linker, referred to as the Drug Affinity Complex, is designed to react with and form a stable thioether bond with Cys34 of circulating serum albumin upon subcutaneous injection. The resulting albumin-peptide conjugate dramatically extends the compound's plasma half-life from approximately 30 minutes (for the unconjugated base peptide) to approximately 6-8 days, enabling sustained stimulation of pituitary growth hormone (GH) release from a single injection.
The base peptide sequence of CJC-1295 incorporates four amino acid substitutions relative to native GHRH(1-29) that confer resistance to dipeptidyl peptidase-IV (DPP-IV) enzymatic degradation: D-Ala at position 2, Gln at position 8, Ala at position 15, and Leu at position 27. These modifications alone extend the half-life compared to native GHRH, but the addition of the DAC moiety produces a qualitatively different pharmacokinetic and pharmacodynamic profile — one characterized by sustained, non-pulsatile elevation of GH and insulin-like growth factor-1 (IGF-1) levels over the course of a week or longer.
Clinical research, most notably the pivotal study by Teichman et al. (2006) published in the Journal of Clinical Endocrinology and Metabolism, demonstrated that a single subcutaneous injection of CJC-1295 with DAC produced dose-dependent increases in GH and IGF-1 that persisted for 6-14 days, with IGF-1 levels remaining elevated for up to 28 days at higher doses. This sustained GH elevation represents both the compound's primary advantage (dosing convenience, consistent anabolic signaling) and its principal limitation (loss of the physiological pulsatile GH secretory pattern).
CJC-1295 with DAC (CAS: 863288-34-0 for the base peptide) remains an important research tool for studying the consequences of sustained versus pulsatile GH axis stimulation, the pharmacology of albumin-binding prodrug strategies, and the physiological roles of GHRH signaling in metabolic regulation, body composition, and tissue repair.
Interactive 3D Molecular Structure
The following interactive 3D visualization renders the CJC-1295 with DAC peptide backbone, highlighting the D-Ala2 substitution and the DAC (maleimidopropionic acid) moiety extending from Lys30 that enables covalent albumin binding in vivo.
Legend: The interactive visualization above depicts the 30-residue alpha-helical backbone of CJC-1295. D-Ala at position 2 is highlighted in purple, indicating the key DPP-IV resistance substitution. The dashed orange chain extending from Lys30 represents the Drug Affinity Complex (DAC) — the maleimidopropionic acid (MPA) linker that covalently binds serum albumin in vivo, extending the peptide's half-life to approximately 6-8 days. Drag to rotate the structure; scroll to zoom.
Table of Contents
- Introduction and Development History
- Molecular Structure and Chemistry
- The Drug Affinity Complex (DAC) Technology
- Mechanism of Action
- Pulsatile vs. Sustained GH Stimulation
- Clinical Research and Pharmacokinetic Studies
- IGF-1 and Body Composition Research
- Safety Profile and Tolerability
- Comparison with Related GHRH Analogs and GH Secretagogues
- Research Applications
- References
- Disclaimer
Introduction and Development History
ConjuChem Biotechnologies and the DAC Platform
CJC-1295 with DAC was developed by ConjuChem Biotechnologies, a Montreal, Canada-based pharmaceutical company specializing in long-acting peptide therapeutics. The company's proprietary Drug Affinity Complex (DAC) technology platform was designed to overcome one of the most significant challenges in peptide pharmacology: the extremely short circulating half-life of native peptide hormones. Native GHRH(1-44) has a plasma half-life of less than 7 minutes due to rapid enzymatic degradation, primarily by dipeptidyl peptidase-IV (DPP-IV), which cleaves the N-terminal Tyr-Ala dipeptide and inactivates the hormone [1].
ConjuChem's strategy was twofold. First, the base peptide was engineered with four amino acid substitutions to resist DPP-IV degradation (producing the modified GHRH analog designated CJC-1295). Second, a reactive maleimidopropionic acid (MPA) moiety was conjugated to the C-terminal lysine residue via a lysine spacer, creating a prodrug that would covalently bind to circulating serum albumin following injection. The albumin-conjugated peptide would then be protected from renal clearance and further proteolysis, dramatically extending its functional half-life [2].
Development Timeline
The compound progressed through preclinical development in the early 2000s and entered human clinical trials under the designation CJC-1295. The landmark Phase I/II dose-escalation study was published by Teichman et al. in 2006 in the Journal of Clinical Endocrinology and Metabolism, establishing the compound's pharmacokinetic and pharmacodynamic profile in healthy adults [3]. ConjuChem also explored the DAC platform for other peptides, including GLP-1 analogs for diabetes, but the GHRH application received the most extensive clinical characterization.
Naming Conventions
It is important to note that in the research literature and peptide marketplace, "CJC-1295" can refer to either the DAC-conjugated form or the unconjugated base peptide (sometimes called "CJC-1295 without DAC" or "modified GRF 1-29"). These two compounds have fundamentally different pharmacokinetic profiles. Throughout this guide, "CJC-1295 with DAC" refers specifically to the albumin-binding, long-acting formulation, while the unconjugated form is referred to as "CJC-1295 without DAC" or "mod GRF 1-29."
Molecular Structure and Chemistry
Base Peptide Sequence
CJC-1295 with DAC is built upon a 30-amino acid GHRH analog with the following sequence:
Tyr-D-Ala-Asp-Ala-Ile-Phe-Thr-Gln-Ser-Tyr-Arg-Lys-Val-Leu-Ala-Gln-Leu-Ser-Ala-Arg-Lys-Leu-Leu-Gln-Asp-Ile-Leu-Ser-Arg-Lys(MPA-Lys)-NH2
The four critical amino acid substitutions relative to native GHRH(1-29)-NH2 are:
- Position 2: Ala replaced with D-Ala — confers resistance to DPP-IV cleavage at the N-terminus
- Position 8: Asn replaced with Gln — prevents asparagine deamidation, improving chemical stability
- Position 15: Gly replaced with Ala — enhances alpha-helical propensity and receptor binding affinity
- Position 27: Met replaced with Leu — eliminates methionine oxidation and improves stability
These modifications collectively produce a GHRH analog with substantially improved metabolic stability while retaining full agonist activity at the GHRH receptor (GHRHR) [4].
The MPA-Lys DAC Moiety
The Drug Affinity Complex is attached at Lys30 and consists of:
- A lysine spacer bridging the peptide backbone to the reactive group
- A maleimidopropionic acid (MPA) moiety that serves as the albumin-reactive element
The maleimide ring of MPA undergoes a rapid and selective Michael addition reaction with the free thiol group of Cys34 on human serum albumin, forming a stable thioether bond. This reaction occurs spontaneously at physiological pH and temperature within minutes of subcutaneous injection [2].
Physicochemical Properties
| Property | Value |
|---|---|
| Base Peptide CAS Number | 863288-34-0 |
| Sequence Length | 30 amino acids + DAC moiety |
| Molecular Weight (base peptide) | Approximately 3,367.9 Da |
| Molecular Weight (with DAC) | Approximately 3,647.3 Da |
| C-terminal | Amidated (-NH2) |
| Appearance | White to off-white lyophilized powder |
| Solubility | Soluble in sterile water, saline, dilute acetic acid |
| Storage | -20C lyophilized; 2-8C reconstituted (use within 21 days) |
| Key Modifications | D-Ala2, Gln8, Ala15, Leu27, Lys(MPA-Lys)30 |
The Drug Affinity Complex (DAC) Technology
Mechanism of Albumin Conjugation
The DAC technology represents an elegant prodrug approach to extending peptide half-life. The process occurs in three stages:
- Injection: CJC-1295 with DAC is administered subcutaneously as a free peptide bearing the reactive MPA group
- Albumin capture: Within minutes of entering the bloodstream, the MPA maleimide ring reacts with the Cys34 thiol on circulating human serum albumin (HSA), forming a covalent thioether bond
- Circulation as a macromolecular conjugate: The resulting approximately 70 kDa albumin-peptide complex circulates with the pharmacokinetic properties of albumin (half-life approximately 19 days), protected from renal filtration and most proteolytic degradation [2, 5]
Why Albumin Binding Extends Half-Life
Human serum albumin possesses several properties that make it an ideal carrier:
- Size: At approximately 66.5 kDa, albumin exceeds the renal filtration threshold (approximately 60 kDa), preventing glomerular clearance
- FcRn recycling: Albumin is recycled by the neonatal Fc receptor (FcRn) in endothelial cells, the same receptor that extends IgG antibody half-life, contributing to its approximately 19-day plasma half-life
- Abundance: Present at 35-50 g/L in human plasma, albumin provides an essentially unlimited supply of Cys34 binding sites
- Protease shielding: The bulky albumin molecule sterically protects the conjugated peptide from circulating proteases [5]
Practical Half-Life versus Albumin Half-Life
While albumin itself has a half-life of approximately 19 days, the functional half-life of CJC-1295 with DAC is approximately 6-8 days. This difference reflects several factors: gradual loss of peptide biological activity while still conjugated, slow hydrolysis of the thioether bond under physiological conditions, and receptor-mediated internalization and degradation of the GHRH analog portion following receptor binding [3].
Mechanism of Action
GHRH Receptor Signaling
CJC-1295 with DAC exerts its pharmacological effects through binding to and activation of the growth hormone-releasing hormone receptor (GHRHR), a G-protein coupled receptor (GPCR) expressed primarily on somatotroph cells of the anterior pituitary gland. The signaling cascade proceeds as follows:
- Receptor binding: The albumin-conjugated CJC-1295 binds to the GHRHR extracellular domain with an affinity comparable to native GHRH
- G-protein activation: GHRHR couples to Gs, activating adenylyl cyclase and increasing intracellular cAMP
- PKA signaling: Elevated cAMP activates protein kinase A (PKA), which phosphorylates multiple downstream targets
- GH gene transcription and secretion: PKA activation promotes both immediate GH secretion from preformed secretory granules and longer-term transcriptional upregulation of the GH1 gene via the Pit-1 transcription factor
- Somatotroph proliferation: Chronic GHRH receptor stimulation promotes somatotroph proliferation and maintains the pituitary GH-secreting cell population [6]
Sustained versus Pulsatile Receptor Activation
A critical distinction between CJC-1295 with DAC and the unconjugated peptide (CJC-1295 without DAC) is the temporal pattern of receptor activation. Native GHRH is released from the hypothalamus in discrete pulses, primarily during slow-wave sleep, producing corresponding pulses of GH secretion. CJC-1295 without DAC, with its approximately 30-minute half-life, mimics this pulsatile pattern when administered as a bolus injection. In contrast, CJC-1295 with DAC produces continuous receptor stimulation over days, resulting in a sustained elevation of baseline GH levels with blunted pulsatility [3, 7].
Interaction with Somatostatin
Even during sustained GHRH receptor stimulation by CJC-1295 with DAC, somatostatin (SRIF) continues to exert periodic inhibitory effects on GH secretion. This means that GH levels under DAC stimulation are not completely flat but show attenuated pulsatile variations superimposed on an elevated baseline. The somatostatin-GHRH interplay partially preserves some degree of rhythmicity, though the amplitude and pattern differ substantially from normal physiology [7, 8].
Pulsatile vs. Sustained GH Stimulation
The Physiological Importance of GH Pulsatility
One of the most actively debated topics in GH biology is the significance of pulsatile versus continuous GH exposure. In normal physiology, GH is secreted in approximately 6-12 discrete pulses per day, with the largest pulses occurring during slow-wave sleep. Research has demonstrated that pulsatile and continuous GH exposure produce qualitatively different biological effects:
- Hepatic gene expression: Pulsatile GH activates a distinct set of liver genes compared to continuous GH. In rodent models, the sexually dimorphic pattern of liver gene expression (including CYP enzymes, MUPs, and growth-related genes) is determined by GH pulse amplitude and frequency rather than total GH exposure [9]
- IGF-1 production: Both pulsatile and continuous GH stimulate hepatic IGF-1 production, but the temporal pattern may influence IGF-1 bioavailability and binding protein expression
- Lipolysis: Pulsatile GH appears to be a more potent stimulus for lipolysis than continuous exposure at equivalent total GH concentrations, potentially through differential activation of the JAK2-STAT5b pathway [10]
- Body composition: Some evidence suggests that pulsatile GH favors lean mass accretion, while continuous GH may promote more uniform tissue growth
Implications for CJC-1295 with DAC
The sustained GH elevation produced by CJC-1295 with DAC may therefore have biological consequences that differ from those of pulsatile GH stimulation achieved with shorter-acting GHRH analogs or GH secretagogues. Researchers studying this compound must consider that:
- Total GH exposure is increased: The area under the GH concentration-time curve (AUC) is significantly greater with DAC versus without DAC
- Peak-to-trough ratio is reduced: The pulsatile pattern is dampened, which may affect downstream signaling
- Feedback dynamics are altered: Sustained GH elevation chronically activates negative feedback through IGF-1 and somatostatin, which may lead to partial desensitization over time
This trade-off between dosing convenience and physiological fidelity is a central consideration in GHRH analog research and represents an important area of ongoing investigation [7, 8].
Clinical Research and Pharmacokinetic Studies
The Teichman et al. (2006) Pivotal Study
The most comprehensive published clinical characterization of CJC-1295 with DAC comes from the study by Teichman et al. published in the Journal of Clinical Endocrinology and Metabolism [3]. This dose-escalation study enrolled healthy adults aged 21-61 years and administered single subcutaneous injections at doses of 30, 60, 90, 120, 150, 200, and 250 mcg/kg.
Key pharmacokinetic findings:
- Plasma CJC-1295 concentrations: Dose-proportional Cmax values were observed, with measurable plasma levels persisting for 6-14 days depending on dose
- Half-life: The estimated elimination half-life was 5.8 to 8.1 days across dose groups
- Albumin binding: Greater than 90% of circulating CJC-1295 was albumin-bound within 15 minutes of injection
Key pharmacodynamic findings:
- GH elevation: Mean GH levels increased 2- to 10-fold above baseline within 1-4 hours of injection and remained elevated for 6-8 days
- IGF-1 elevation: Serum IGF-1 levels increased 1.5- to 3-fold above baseline, with peak levels occurring at days 3-7 and remaining elevated for up to 14-28 days at higher doses
- IGFBP-3: Insulin-like growth factor binding protein-3 levels also increased in a dose-dependent manner
- Dose-response: Clear dose-dependent increases in both GH and IGF-1 AUC values were observed across the dose range
Multiple-Dose Studies
Subsequent research examined weekly dosing of CJC-1295 with DAC over 2-4 weeks. Alba et al. (2006) reported on the effects of weekly subcutaneous injections (30 and 60 mcg/kg) for 2-4 weeks in healthy adults [11]. Key findings included:
- Cumulative IGF-1 elevation: Weekly dosing produced progressive increases in IGF-1 levels, reaching a plateau approximately 2-3 weeks after initiation
- Sustained GH elevation: Trough GH levels remained above baseline throughout the dosing period, confirming sustained (non-pulsatile) GH stimulation
- No tachyphylaxis: GH and IGF-1 responses did not diminish with repeated dosing over the study period, suggesting absence of receptor desensitization at these doses and durations
Pharmacokinetic Modeling
Population pharmacokinetic analysis of the clinical data yielded a two-compartment model with first-order absorption and elimination. The albumin-binding step was modeled as a rapid, essentially irreversible process. Key modeled parameters included [3, 11]:
| Parameter | Value |
|---|---|
| tmax (plasma) | 2-4 hours |
| Elimination half-life | Approximately 6-8 days |
| Volume of distribution | Approximately 70-80 mL/kg (albumin space) |
| Bioavailability (SC) | Approximately 85-95% (albumin-bound form) |
| GH peak elevation | 2-10x baseline (dose-dependent) |
| IGF-1 peak elevation | 1.5-3x baseline (dose-dependent) |
| Duration of IGF-1 elevation | 14-28 days (dose-dependent) |
IGF-1 and Body Composition Research
IGF-1 Axis Modulation
The sustained elevation of IGF-1 produced by CJC-1295 with DAC has generated significant research interest due to the pleiotropic roles of IGF-1 in physiology:
- Anabolic signaling: IGF-1 activates the PI3K/Akt/mTOR pathway, promoting protein synthesis in skeletal muscle and other tissues
- Anti-catabolic effects: IGF-1 suppresses protein degradation pathways, including the ubiquitin-proteasome system and autophagy
- Metabolic regulation: IGF-1 enhances glucose uptake and utilization, and modulates lipid metabolism
- Tissue repair: IGF-1 promotes wound healing, bone formation, and connective tissue remodeling [12]
The sustained IGF-1 elevation from CJC-1295 with DAC differs from that produced by exogenous GH administration in that it preserves the GH-to-IGF-1 physiological ratio and maintains concurrent elevation of IGFBP-3, which modulates IGF-1 bioactivity. This distinction is relevant for researchers studying the differential effects of "GH-driven" versus "direct" IGF-1 elevation [3].
Preclinical Body Composition Data
Animal studies using GHRH analogs with extended half-lives have demonstrated dose-dependent effects on body composition, including increased lean body mass, reduced adiposity, and improved bone mineral density. While published preclinical data specific to CJC-1295 with DAC is limited in the public domain (much of ConjuChem's preclinical data remains proprietary), studies with comparable long-acting GHRH analogs have shown [13, 14]:
- Increased nitrogen retention and protein synthesis rates in skeletal muscle
- Reduced visceral and subcutaneous adipose tissue mass
- Enhanced bone mineral content and trabecular bone volume
- Improved collagen synthesis and wound healing rates
Safety Profile and Tolerability
Clinical Trial Safety Data
Across published clinical studies, CJC-1295 with DAC demonstrated a generally favorable safety profile at doses up to 250 mcg/kg [3, 11]:
- Injection site reactions: Mild, transient erythema and induration at the injection site were the most common adverse events, reported in approximately 15-25% of subjects
- GH-related side effects: Transient fluid retention, joint stiffness, and paresthesias were reported at higher doses, consistent with elevated GH/IGF-1 levels
- Metabolic parameters: Fasting glucose and insulin levels were not significantly altered, though some subjects showed transient glucose elevation within the first 24-48 hours
- Cortisol and prolactin: No clinically significant changes in cortisol, prolactin, or thyroid hormone levels were observed
- Antibody formation: No anti-CJC-1295 or anti-GH antibodies were detected
Theoretical Safety Considerations
Researchers should be aware of several theoretical concerns associated with sustained GH axis stimulation:
- Prolonged IGF-1 elevation: Chronic elevation of IGF-1 above the physiological range has been epidemiologically associated with increased risk of certain malignancies, though the clinical significance at the levels produced by CJC-1295 with DAC is not established [15]
- Loss of pulsatility: The consequences of chronic non-pulsatile GH stimulation on hepatic function, glucose metabolism, and other GH-dependent processes remain incompletely characterized
- Somatotroph hyperplasia: Theoretical risk of pituitary somatotroph hyperplasia with very long-term, continuous GHRH stimulation, though this has not been observed in clinical studies of limited duration
Comparison with Related GHRH Analogs and GH Secretagogues
The following table provides a comparative overview of CJC-1295 with DAC relative to related research compounds:
| Feature | CJC-1295 with DAC | CJC-1295 without DAC (mod GRF 1-29) | Sermorelin (GRF 1-29) | Tesamorelin (GHRH analog) |
|---|---|---|---|---|
| Structure | 30 AA + DAC (MPA-Lys) | 30 AA (same base) | 29 AA (native GHRH 1-29) | 44 AA (trans-3-hexenoic acid-GHRH 1-44) |
| Half-life | Approximately 6-8 days | Approximately 30 minutes | Approximately 10-12 minutes | Approximately 26 minutes |
| Dosing frequency | Weekly | 2-3x daily (research) | 2-3x daily (research) | Daily |
| GH stimulation pattern | Sustained (non-pulsatile) | Pulsatile (bolus-dependent) | Pulsatile (bolus-dependent) | Pulsatile (bolus-dependent) |
| IGF-1 elevation | Sustained (1.5-3x baseline, weeks) | Transient (hours) | Transient (hours) | Moderate sustained (daily dosing) |
| DPP-IV resistance | Yes (D-Ala2) | Yes (D-Ala2) | No (rapidly degraded) | Partial |
| Albumin binding | Yes (covalent) | No | No | No |
| FDA-approved | No | No | Yes (discontinued) | Yes (HIV lipodystrophy) |
| Primary research use | Sustained GH/IGF-1 elevation | Pulsatile GH release studies | GH deficiency research | Visceral fat reduction |
| Developer | ConjuChem Biotechnologies | Derived from ConjuChem work | Serono (EMD Serono) | Theratechnologies |
Key Differentiators
CJC-1295 with DAC vs. CJC-1295 without DAC: These compounds share an identical base peptide sequence but produce fundamentally different pharmacodynamic profiles. The DAC version provides sustained, non-pulsatile GH stimulation from weekly dosing, while the non-DAC version produces discrete GH pulses resembling normal physiology. The choice between them depends entirely on the research question: sustained GH/IGF-1 elevation studies favor the DAC version, while pulsatile GH physiology studies favor the non-DAC form [3, 7].
CJC-1295 with DAC vs. Sermorelin: Sermorelin is the native GHRH(1-29) sequence without any protective modifications. It is rapidly degraded by DPP-IV and has a half-life of only approximately 10-12 minutes. While Sermorelin was historically approved for GH deficiency diagnosis and treatment, its extremely short duration of action limits its utility for sustained GH axis research [16].
CJC-1295 with DAC vs. Tesamorelin: Tesamorelin is an FDA-approved GHRH analog (for HIV-associated lipodystrophy) with a trans-3-hexenoic acid modification that improves stability. It has a half-life of approximately 26 minutes and requires daily injection. Tesamorelin produces pulsatile GH stimulation and has the most extensive clinical safety database among GHRH analogs [17].
Research Applications
CJC-1295 with DAC serves as a valuable tool across multiple research domains:
GH Axis Pharmacology
- Studying the pharmacological consequences of sustained versus pulsatile GHRH receptor activation
- Investigating GHRH receptor desensitization kinetics under continuous stimulation
- Characterizing the somatostatin-GHRH interaction during chronic GHRH exposure
- Modeling long-acting peptide-albumin conjugate pharmacokinetics
Metabolic and Body Composition Research
- Examining the effects of sustained GH/IGF-1 elevation on protein synthesis and nitrogen balance
- Investigating differential effects of pulsatile versus continuous GH on lipolysis and lipogenesis
- Studying the metabolic consequences of chronic IGF-1 elevation at varying levels
- Comparing body composition outcomes between sustained and pulsatile GH stimulation paradigms
Drug Delivery Technology
- Validating albumin-binding prodrug approaches for peptide half-life extension
- Studying maleimide-thiol conjugation chemistry in vivo
- Developing pharmacokinetic models for albumin-conjugated therapeutics
- Informing the design of next-generation long-acting peptide drugs
Combination Research
CJC-1295 with DAC has been studied in combination with growth hormone secretagogues (GHS), particularly ghrelin mimetics such as Ipamorelin and GHRP-6. The combination of a GHRH analog with a GHS produces synergistic GH release, as these compounds act through distinct receptor systems (GHRHR versus GHS-R1a) and signaling pathways. Researchers have explored whether combining sustained GHRH stimulation (via DAC) with pulsatile GHS dosing might partially restore physiological GH pulsatility while maintaining elevated baseline GH levels [18].
Related Peptides of Interest
Researchers studying CJC-1295 with DAC may also wish to investigate:
- CJC-1295 without DAC (mod GRF 1-29): The same base peptide without albumin binding, for pulsatile GH studies
- Ipamorelin: A selective ghrelin receptor agonist that produces clean GH pulses without cortisol or prolactin effects
- Sermorelin: The native GHRH(1-29) fragment for comparative studies
References
[1] Frohman, L.A., Downs, T.R., & Chomczynski, P. (1992). "Regulation of growth hormone secretion." Frontiers in Neuroendocrinology, 13(4), 344-405.
[2] Bhatt, R., de Vries, P., Tulinsky, J., et al. (2003). "Synthesis and in vivo evaluation of a long-acting human growth hormone-releasing factor analog using drug affinity complex (DAC) technology." Peptides for Youth, 59-60. DOI: 10.1007/978-0-387-73657-0_26
[3] Teichman, S.L., Neale, A., Lawrence, B., Gagnon, C., Castaigne, J.P., & Bhatt, R. (2006). "Prolonged stimulation of growth hormone (GH) and insulin-like growth factor I secretion by CJC-1295, a long-acting analog of GH-releasing hormone, in healthy adults." Journal of Clinical Endocrinology and Metabolism, 91(3), 799-805. DOI: 10.1210/jc.2005-1536
[4] Izdebski, J., Pinski, J., Horvath, J.E., Halmos, G., Nagy, A., & Schally, A.V. (1995). "Synthesis and biological evaluation of superactive agonists of growth hormone-releasing hormone." Proceedings of the National Academy of Sciences, 92(10), 4872-4876. DOI: 10.1073/pnas.92.10.4872
[5] Sleep, D., Cameron, J., & Evans, L.R. (2013). "Albumin as a versatile platform for drug half-life extension." Biochimica et Biophysica Acta, 1830(12), 5526-5534. DOI: 10.1016/j.bbagen.2013.04.023
[6] Mayo, K.E., Miller, T.L., DeAlmeida, V., et al. (2000). "Regulation of the pituitary somatotroph cell by GHRH and its receptor." Recent Progress in Hormone Research, 55, 237-266.
[7] Ionescu, M. & Bhatt, R. (2006). "Pharmacokinetic profile of CJC-1295, a long-acting growth hormone-releasing hormone analog." Endocrine Reviews, 27(S1), P2-592.
[8] Veldhuis, J.D. & Bowers, C.Y. (2010). "Integrating GHS into the ghrelin system." International Journal of Peptides, 2010, 879503. DOI: 10.1155/2010/879503
[9] Waxman, D.J. & O'Connor, C. (2006). "Growth hormone regulation of sex-dependent liver gene expression." Molecular Endocrinology, 20(11), 2613-2629. DOI: 10.1210/me.2006-0007
[10] Nordstrom, S.M., Tran, J.L., Sos, B.C., Wagner, K.U., & Bhatt, R. (2011). "Disruption of JAK2 in adipocytes impairs lipolysis and improves fatty liver in mice with elevated GH." Molecular Endocrinology, 25(11), 1863-1876. DOI: 10.1210/me.2011-1110
[11] Alba, M., Fintini, D., Sagazio, A., et al. (2006). "Once-daily administration of CJC-1295, a long-acting growth hormone-releasing hormone (GHRH) analog, normalizes growth in the GHRH knockout (GHRHKO) mouse." American Journal of Physiology-Endocrinology and Metabolism, 291(6), E1290-E1294. DOI: 10.1152/ajpendo.00172.2006
[12] Laron, Z. (2001). "Insulin-like growth factor 1 (IGF-1): a growth hormone." Journal of Clinical Pathology: Molecular Pathology, 54(5), 311-316. DOI: 10.1136/mp.54.5.311
[13] Clark, R., Olson, K., Fuh, G., et al. (1996). "Long-acting growth hormones produced by conjugation with polyethylene glycol." Journal of Biological Chemistry, 271(36), 21969-21977. DOI: 10.1074/jbc.271.36.21969
[14] Svensson, J., Lall, S., Dickson, S.L., et al. (2000). "The GH secretagogues ipamorelin and GH-releasing peptide-6 increase bone mineral content in adult female rats." Journal of Endocrinology, 165(3), 569-577. DOI: 10.1677/joe.0.1650569
[15] Renehan, A.G., Zwahlen, M., Minder, C., O'Dwyer, S.T., Shalet, S.M., & Egger, M. (2004). "Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis." Lancet, 363(9418), 1346-1353. DOI: 10.1016/S0140-6736(04)16044-3
[16] Walker, R.F., Codd, E.E., Barone, F.C., et al. (1990). "Oral activity of the growth hormone releasing peptide His-D-Trp-Ala-Trp-D-Phe-Lys-NH2 in rats, dogs and monkeys." Life Sciences, 47(1), 29-36. DOI: 10.1016/0024-3205(90)90024-L
[17] Falutz, J., Allas, S., Blot, K., et al. (2007). "Metabolic effects of a growth hormone-releasing factor in patients with HIV." New England Journal of Medicine, 357(23), 2359-2370. DOI: 10.1056/NEJMoa072375
[18] Bowers, C.Y. (2012). "Growth hormone-releasing peptide (GHRP)." Cellular and Molecular Life Sciences, 55(8-9), 1426-1439. DOI: 10.1007/s000180050383
Disclaimer
This product description is intended for informational and research purposes only. CJC-1295 with DAC is sold as a research peptide and is not intended for human consumption, therapeutic use, or as a dietary supplement. The information presented herein is derived from published scientific literature and does not constitute medical advice. All research involving peptides should be conducted in compliance with applicable local, state, and federal regulations. Researchers should consult relevant institutional review boards and regulatory bodies before initiating any research protocols.
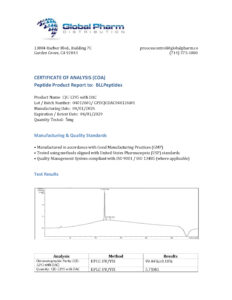
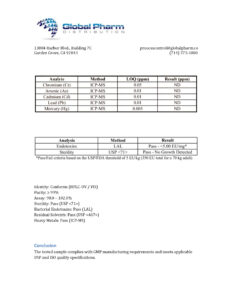
BLL Peptides provides research-grade peptides for qualified researchers and institutions. Product purity is verified by HPLC and mass spectrometry analysis. Certificates of analysis are available upon request.
CJC-1295 with DAC is a research-grade extended-release GHRH analog using Drug Affinity Complex technology, studied for prolonged half-life, sustained GH and IGF-1 elevation, and body composition research. Researchers investigating long-duration growth hormone stimulation rely on pharmaceutical-grade purity for valid outcomes. Available at BLL Peptides — USA-made, rigorously tested.
| ✅ COA tested every batch | ✅ 98%+ purity guaranteed |
| ✅ USA manufactured, GMP-certified | ✅ Glass vials — not plastic |
| ✅ Veteran-owned company | ✅ Free shipping over $150 |